Global Stem Cells Group announces participation in IV Jornadas de Actualización Medestética 2023
Global Stem Cells Group (GSCG), a leading provider of regenerative medicine solutions, is proud to announce its participation in the IV Jornadas de Actualización Medestética 2023 through the International Society for Stem Cell Association (ISSCA), its educational division and also with its line of Cellgenic Exosomes products.
The congress, which will take place on March 30th and 31st at Paseo La Plaza in Buenos Aires, Argentina, will provide a platform for ISSCA to share its knowledge and expertise in regenerative medicine with the Argentine medical community.
ISSCA is a multidisciplinary community of physicians and scientists whose mission is to advance the science, technology, and practice of regenerative medicine to treat disease and lessen human suffering. The organization has been at the forefront of promoting excellent and standardized regenerative medicine and will continue to participate in similar medical events such as this one.
The Global Stem Cells Group will also have a booth at the event to showcase its products, Cellgenic Exosomes, in its two presentations: “Cellgenic Lyophilized Exosomes” and “Cellgenic Flow Exosomes.” Additionally, it will provide guidance on how to use the products on patients.
As part of the program, ISSCA faculty members will be giving presentations and holding practical workshops to demonstrate the latest applications of regenerative medicine. The ISSCA program will cover all aspects of regenerative medicine including guidelines and clinical protocols.
“We are excited to take part in this congress and share our expertise, knowledge, and cellgenic exosome product line with the Argentine medical community,” said Benito Novas, Managing Director of Global Stem Cells Group and Head of Public Relations for the International Society for Stem Cell Application from the USA.
The IV Jornadas de Actualización Medestética 2023 will feature various ISSCA-certified doctors and experts as speakers, including:
Dr. Silvina Pastrana
Dr. Pastrana heads the ISSCA chapter in Argentina and serves as the medical center’s director. “Traditional, advanced, and cutting-edge anti-aging and rejuvenation medicine” will be her theme at this congress. Dr. Silvina will deliver a 40-minute summary of all the regenerative medicine and cell therapies that are now being used to postpone aging and enhance quality of life. therapies using NK cells, fluid and lyophilized exosomes, allogenic therapies, and autologous cells.
Dr. Andrea Lapeire
Dr. Lapeire is a doctor of aesthetic medicine with advanced degrees in environment, homotoxicology, biological medicine, and obesity. She will speak on alopecias: traditional and cutting-edge. Exosome Use and Biological Stimulation Fundamentals.
Prof. Dr. Julio Ferreira
Professor Ferreira is from Argentina and is a professional cosmetic surgery and aesthetic medicine expert. He is a university professor of medicine and is the president of the South American Academy of Aesthetic Surgery. He will cover regenerative therapies in rejuvenation and facial harmonization in his lecture.
Dr. Alfonso Ciancaglini
A specialist in Medical Clinic and regenerative medicine, Dr. Alfonsoi is a member of the International Society For Cellular Therapy Application and holds a certification from the Stem Cells Group. His presentation will discuss the use of regenerative medicine to treat venous ulcers.
Benito Novas
Benito Novas is the Head of Public Relations for the International Society for Stem Cell Application from the USA. He is the senior marketing consultant and founder of “Estética marketing INC” as an Author, he has authored several books among them: Su clinica este’tica. Lo que dicen sus pacientes” Marketing Digital en su Clinica Estetica” and “Marketing en Tiempos de Crisis” for more information on Novas you can check on www.benitonovas.com. In this congress. Benito Novas will be covering Digital Marketing in Cash-based practices.
ISSCA at Global Perspective.
ISSCA has been at the forefront of promoting excellent and standardized regenerative medicine. For more information and updates. visit us at https://www.issca.us/
The ISSCA is a multidisciplinary community of physicians and scientists with a mission to advance the science, technology, and practice of Regenerative Medicine to treat disease and lessen human suffering. Its members are leaders in setting standards and promoting excellence in regenerative medicine, related education, certification, research and publications.
About Global Stem Cell Group
The Global Stem Cell Group is a family of several companies focused on stem cell medicine and research. The company uses its network to bring leadership in regenerative medicine training, research, and patient applications.
GSCG’s mission is to allow physicians to present the benefits of stem cell medicine to patients worldwide. The company also partners with policymakers, educators, and regulators to promote regenerative medicine.
Global Stem Cells Group is a publicly traded company operating under the symbol MSSV. https://finance.yahoo.com/quote/mssv/
To learn more about Global Stem Cells Group, Inc.’s companies visit our website www.stemcellsgroup.com or call +1 305 560 5331
Safe Harbor Statement: Statements in this news release may be “forward-looking statements”. Forward-looking statements include, but are not limited to, statements that express our intentions, beliefs, expectations, strategies, predictions, or any other information relating to our future activities or other future events or conditions. These statements are based on current expectations, estimates, and projections about our business based partly on assumptions made by management. These statements are not guarantees of future performance and involve risks, uncertainties, and assumptions that are difficult to predict. Therefore, actual outcomes and results may and are likely to differ materially from what is expressed or forecasted in forward-looking statements due to numerous factors. Any forward-looking statements speak only as of the date of this news release, and The Global Stem Cells Group undertakes no obligation to update any forward-looking statement to reflect events or circumstances after the date of this news release. This press release does not constitute a public offer of any securities for sale. Any securities offered privately will not be or have not been registered under the Act and may not be offered or sold in the United States absent registration or an applicable exemption from registration requirements.
- Published in Press Releases
Clinical trials on NK cells give hope for many people who are suffering from cancer.
Induced Pluripotent Stem Cells (iPSCs)
Induced pluripotent stem cells (also known as iPS cells or iPSCs) are a type of pluripotent stem cell that can be generated directly from a somatic cell. Pluripotent stem cells hold promise in the field of regenerative medicine. Because they can propagate indefinitely, as well as give rise to every other cell type in the body (such as neurons, heart, pancreatic, and liver cells), they represent a single source of cells that could be used to replace those lost to damage or disease.
Natural Killer Cells and Their Role
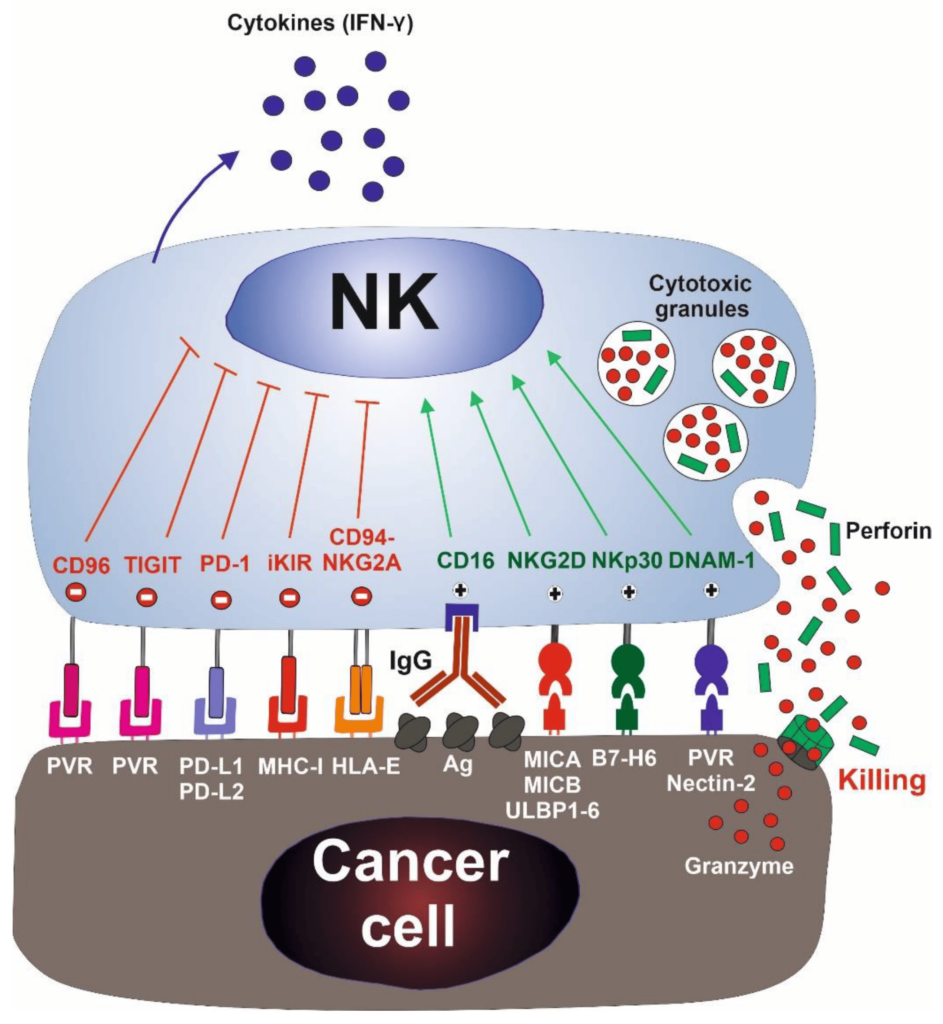
Natural killer (NK) cells are a type of cytotoxic lymphocyte critical to the innate immune system. The role NK cells play is analogous to that of cytotoxic T cells in the vertebrate adaptive immune response. NK cells provide rapid responses to virus-infected cells, acting at around three days after infection, and respond to tumor formation.
Typically, immune cells detect the major histocompatibility complex (MHC) presented on infected cell surfaces, triggering cytokine release and causing apoptosis. NK cells are unique, however, as they can recognize stressed cells in the absence of antibodies and MHC, allowing for a much faster immune reaction.
Clinical Trials on NK Cells
Overview of NK Cell Clinical Trials
In a first clinical trial, a natural killer cell immunotherapy derived from induced pluripotent stem cells is being tested for safety in 64 patients with a variety of solid tumors. The first subjects used for the study received the cells in February at the University of California, San Diego (UCSD) Moores Cancer Center and MD Anderson Cancer Center.
Targeted Cancer Types
This study targets late-stage cancer patients with solid tumors, including lymphoma, colorectal cancer, and breast cancer. The FT500 NK cells do not undergo any further alterations after their derivation from induced pluripotent stem cells (iPSCs), offering the possibility of a quicker, ready-made treatment.
Human Embryonic Stem Cells and iPSCs
Human embryonic stem cells (hESCs) and induced pluripotent stem cells (iPSCs) provide an accessible, genetically tractable, and homogenous starting cell population to efficiently study human blood cell development. These cell populations provide platforms to develop new cell-based therapies to treat both malignant and nonmalignant hematological diseases.
NK Cells and Cancer Cell Targeting
The NK cells are immune cells in the same family as T and B cells and are very good at targeting cancer cells for destruction. Laboratory experiments have shown they do so by attacking cells that have lost their significant self-recognition signals that tell the immune system not to attack. This is the phenomenon that can happen among cancer cells but not healthy cells. Experts are not sure how many cancer cells lose that signal. Researchers are hopeful that the clinical trial can help determine which cancer patients could benefit the most from NK cell treatment.
iPS Clones and Tumor Formation
The ability to induce pluripotent stem cells from committed, human somatic cells provides tremendous potential for regenerative medicine. However, there is a defined neoplastic potential inherent to such reprogramming that must be understood and may offer a model for critical understanding of events in the formation of tumors. Using genome-wide assays, we identify cancer-related epigenetic abnormalities that arise early during reprogramming and persist in induced pluripotent stem cell (iPS) clones. These include hundreds of abnormal gene silencing events, patterns of aberrant responses to epigenetic-modifying drugs resembling those for cancer cells, and the presence in iPS and partially reprogrammed cells of cancer-specific gene promoter DNA methylation alterations.
Modification of T Cells for Cancer Therapy
Progress in Adoptive T-cell Therapy
Progress in adoptive T-cell therapy for cancer and infectious diseases is hampered by the lack of readily available antigen-specific human T lymphocytes. Pluripotent stem cells could provide an estimable source of T lymphocytes, but the therapeutic potential of human pluripotent stem cell-derived lymphoid cells generated to date remains uncertain.
CAR T-Cell Therapy
Recently, some approved cell therapies for cancer also rely on modifying T cells, in those cases to produce cancer cell–binding chimeric antigen receptors (CARs), and have been effective in treating certain cancers such as leukemia.
Application of CAR T-Cell Therapy in Solid Tumors
The CAR T technology has wowed the field by all but obliterating some patients’ blood cancers, but solid malignancies present new challenges.
Therapies containing such chimeric antigen receptor (CAR) T cells have been approved for some types of so-called liquid cancers of the blood and bone marrow, such as large B-cell lymphoma and B-cell acute lymphoblastic leukemia. However, the approach has not had as much success for solid tumors.
Challenges and Innovations
Serious research into the therapy for brain cancer started almost 20 years ago after cancer biologist Waldemar Debinski, then at Penn State, discovered that the receptor for the immune signaling molecule interleukin 13 (IL-13) was present on glioblastomas, but not on healthy brain tissue. The receptor thus seemed like an excellent target to home in on cancer cells while sparing healthy ones. The CAR spacer domain that spans the immune cells’ membranes and its intracellular co-stimulatory areas, as well as the process used to expand cells outside the body, boosts the T cells’ activity.
CAR T-Cell Therapy: A Safer Approach
While managing CAR T-cell therapy toxicity could help keep already-designed treatments on their march to the clinic, many immunotherapy companies are also working to develop a new generation of inherently safer therapies, yet just as efficient. A crucial part of achieving this goal will be improving CAR specificity for target cells. With current treatments, the destruction of normal cells is often an unavoidable side effect when healthy tissue carries the same antigens as tumors; noncancerous B cells, for example, are usually casualties in CD19-targeted therapies.
Delivery Challenges
CAR T delivery is a challenging factor in the treatment of solid tumors and other unknown forms of tumors. With non-solid cancers, cells are administered by a blood infusion, and once in circulation, the CAR T cells can seek out and destroy the rogue cells. For solid tumors, it’s not so simple.
Future Directions
The main drawback of taking cells from a patient and developing them into a cellular immunotherapy product is that the process can take weeks.
Patel tells The Scientist, “But for the majority of patients who may not be a candidate or may not have time to wait for such an approach, the idea that there’s off-the-shelf immunotherapy that could potentially act as a living drug against their cancer, I think is a fascinating concept.”
- Published in Corporate News / Blog