ISSCA to Conduct Regenerative Medicine Conference in Cancún, México
Miami, FL, December 19, 2022 – The International Association for Stem Cell Application (ISSCA) has announced plans to host Applications of Stem Cell Therapies in Medicine and Aesthetic Surgery, a regenerative medicine conference, in Cancun, Mexico, April 22, 23, and 24, 2023.
Growing knowledge can turn the tide in the clinical and translational spectrum of stem-cell-based research and therapy, and several obstacles must be overcome. ISSCA’s International Conference, champions new ideas to propel the shift from traditional healthcare to regenerative medicine and therapies. The Conference will also emphasize the role of cutting technology and developments in all areas of stem cell research.
April 22nd & 23rd
The Cancun event will feature a panel of internationally renowned experts on stem cells and regenerative medicine, who will offer a two-day rigorous scientific discourse on critical topics in stem cell research and clinical applications. Topics of focus at the conference include: Tissue engineering and regenerative medicine; updates in Stem Cell research Clinical applications of regenerative medicine and cellular therapies; the latest methods of harvest and isolation, and Non-invasive surgical protocols; strategies for successful delivery of cell products; Ethical and regulatory issues in stem cell technology; and taking advantage of the latest technologies to improve marketing and networking.
April 24th
The third day of the conference (April 24th) will involve intensive hands-on training sessions with ISSCA-certified instructors on stem cell therapy’s aesthetic and clinical applications.
Participants will gain experience and access to different technologies, brands, and products for stem cell therapy.
The leading international conference offers an excellent opportunity for medical practitioners and stakeholders to network and learn from the best in the industry.
All practitioners and stakeholders in the medical sector are invited, including:
- Doctors Researchers & innovators,
- Decision-makers and funding agencies,
- Experts seeking collaborative work on stem cell technology,
- Professional bodies, and
- Marketing professionals.
To learn more about the ISSCA Cancun, Mexico conference and to make a reservation, visit the https://www.issca.us/issca-world-conferences/ website, email info@stemcellsgroup.com, or call +1305 560 5337.
About ISSCA
The International Society for Stem Cell Application (ISSCA) is a multidisciplinary community of scientists and physicians who aspire to treat diseases and lessen human suffering through advances in science, technology, and regenerative medicine.
ISSCA updates its members on advances in stem cell research, MSC, exosomes, and regenerative medicine.
The ISSCA’s vision is to take a leadership position in promoting excellence and setting standards in the regenerative medicine fields of publication, research, education, training, and certification.
The international community provides a platform for practitioners to interact with scientists and build medical networks necessary for marketing.
As a medical specialty, regenerative medicine standards and certifications are essential, which is why ISSCA offers certification training in cities worldwide. The goal is to encourage more physicians to practice regenerative medicine and make it available to patients nationally and internationally. Incorporated under the Republic of Korea as a non-profit company, the ISSCA is focused on fostering excellence and standards in regenerative medicine.
About Global Stem Cells Group
Global Stem Cells Group is the parent company of six companies that are dedicated to stem cell research, solutions, and technology training. The group was founded in 2012 and combines dedicated researchers, patient educators, and physician trainers with the shared goal of meeting the need for high-end stem cell solutions and treatments.
Given that the group has a singular focus in this field, it is uniquely positioned to become the global leader in cellular medicine. In addition, by bringing together leading professionals in cellular medicine, it can discover issues that the industry faces and focus its research and development in this area. This specialization has, undoubtedly, enabled it to come up with solutions that address some of the significant issues that most stakeholders are facing in the industry.
Global Stem Cells Group is a publicly traded company operating under the symbol MSSV. https://finance.yahoo.com/quote/mssv/
- Published in Press Releases
A federal judge recently ruled in favor of a regenerative medicine clinic against the FDA
The FDA brought the lawsuit against Cell Surgical Network Clinic in 2018 in an attempt to assert regulatory authority over stem cell therapies. The agency argued that a physician’s use of his patient’s own stem cells as part of the medical treatment provided to the patient was equivalent to manufacturing a biological drug product and therefore subject to regulation by the FDA.
The court rejected this argument on Tuesday, ruling that the surgical procedure does not create a new prescription drug. The court wrote: “The adipose tissue Defendants remove from patients clearly consists of human cells. And whatever is injected back into patients as part of Defendants’ SVF Surgical Procedure and Expanded MSC Surgical Procedure certainly contains such cells.”
This news opens up the possibilities and sets an important precedent for clinics in the United States to use autologous cellular treatments derived from adipose tissue, giving patients the right to use their own tissue to enhance regenerative processes and fight disease.
“We appreciate the Court’s clear and unequivocal ruling, which affirms what we have been saying for 12 years: that our innovative surgical approach to personal cell therapy is safe and legal,” said Dr. Elliott Lander, co-founder of CSCTC. “With this victory behind us, we look forward to refocusing our energy on our practice and leveraging life-changing stem cell treatments to support physicians and benefit patients across the country.”
As science advances, it’s inevitable that there will be more diverse forms of treatment with human stem cells. Whether the FDA gets on board and expands its rules about what is and is not legal is another question.
Unfortunately, much of what the FDA does falls into the category of politics. It’s usually unwise to make a decision about a particular kind of therapy or medical treatment based on whether it’s technically, currently legal.
It’s helpful to recall that many treatments that are fully legal began as experimental procedures that were not legally offered to the public but only took place in controlled research environments.
The good news is that stem cell therapy continues to make rapid advances on several different fronts, primarily those related to joint pain and associated discomfort. Athletes have made good use of the treatment and were, in fact, among the first major wave of clients for the technique when it was still brand new. After that, word spread about the effectiveness of stem cell treatment as more and more people turned to it as an alternative to surgery and prescription medication.
- Published in Press Releases
Stem cell treatment could offer one-end-solution to Diabetes
Insulin-producing cells grown in the lab could provide a possible cure for the age-long disease (diabetes).
Type 1 diabetes is an auto¬immune disease that wipes out insulin-producing pancreatic beta cells from the body and raises blood glucose to dangerously high levels. These high levels of Blood sugar level can be even fatal. Patients are being administered insulin and given other medications to maintain blood sugar level. To those who cannot maintain their blood sugar level, they are given beta-cell transplants but to tolerate beta cell transplants; patients have to take immunosuppressive drugs as well.
A report by a research group at Harvard University tells us that they used insulin-producing cells derived from human embryonic stem cells (ESCs) and induced pluripotent stem cells to lower blood glucose levels in mice. Nowadays, many laboratories are getting rapid progress in human stem cell technology to develop those cells that are functionally equivalent to beta-cells and the other pancreatic cell types. Other groups are developing novel biomaterials to encapsulate such cells and protect them against the immune system without the need for immunosuppressant.
Major pharmaceutical companies and life sciences venture capital firms have invested more than $100 million in each of the three most prominent biotechnological industries to bring such treatments into clinical use:
- Cambridge
- Massachusetts–based companies Semma Therapeutics
- Sigilon Therapeutics, and ViaCyte of San Diego
Researchers of UC San Francisco have transformed human stem cells into mature insulin-producing cells for the first time, a breakthrough in the effort to develop a cure for type-1 (T1) Diabetes. Replacing these cells, which are lost in patients with T1 diabetes, has long been a dream of regenerative medicine, but until now scientists had not been able to find out how to produce cells in a lab dish that work as they do in healthy adults.
What is T1 diabetes?
T1 diabetes is an autoimmune disorder that destroys the insulin-producing beta cells of the pancreas, typically in childhood. Without insulin’s ability to regulate glucose levels in the blood, spikes in blood sugar can cause severe organ damage and eventually death. The condition can be managed by taking regular shots of insulin with meals. However, people with type 1 diabetes still often experience serious health consequences like kidney failure, heart disease and stroke. Patients facing life-threatening complications of their condition may be eligible for a pancreas transplant from a deceased donor, but these are rare, and they are supposed to wait a long time.
Researchers have just made a breakthrough that might one day make these technologies obsolete, by transforming human stem cells into functional insulin-producing cells (also known as beta cells) – at least in mice.
“We can now generate insulin-producing cells that look and act a lot like the pancreatic beta cells you and I have in our bodies,” explains one of the team, Matthias Hebrok from the University of California San Francisco (UCSF).
“This is a critical step towards our goal of creating cells that could be transplanted into patients with diabetes.”
Type-1 diabetes is characterized by a loss of insulin due to the immune system destroying cells in the pancreas – hence, type 1 diabetics need to introduce their insulin manually. Although this is a pretty good system, it’s not perfect.
Making insulin-producing cells from stem cells
Diabetes can be cured through an entire pancreas transplant or the transplantation of donor cells that produce insulin, but both of these options are limited because they rely on deceased donors. Scientists had already succeeded in turning stem cells into beta cells, but those cells remained stuck at an early stage in their maturity. That meant they weren’t responsive to blood glucose and weren’t able to secrete insulin in the right way.
Scientists at the University of California San Francisco made a breakthrough in the effort to cure diabetes mellitus type 1.
For the first time, researchers transformed human stem cells into mature insulin-producing cells, which could replace those lost in patients with the autoimmune. There is currently no known way to prevent type-1 (T1) diabetes, which destroys insulin production in the pancreas, limits glucose regulation, and results in high blood sugar levels. The condition can be managed with regular shots of insulin, but people with the disease often experience serious health complications like kidney failure, heart disease, and stroke.
“We can now generate insulin-producing cells that look and act a lot like the pancreatic beta cells you and I have in our bodies,” according to Matthias Hebrok, senior author of a study published last week in the journal Nature Cell Biology.
“This is a critical step toward our goal of creating cells that could be transplanted into patients with diabetes,” Hebrok, director of the UCSF Diabetes Center, said in a statement.
Islets of Langerhans are groupings of cells that contain healthy beta cells, among others. As beta cells develop, they have to separate physically from the pancreas to form these islets.
The team artificially separated the pancreatic stem cells and regrouped them into these islet clusters. When they did this, the cells matured rapidly and become responsive to blood sugar. In fact, the islet clusters developed in ways “never before seen” in a lab. After producing these mature cells, the team transplanted them into mice. Within days, the cells were producing insulin similar to the islets in the mice. While the study has been successful in mice, it still needs to go through more rigorous testing to see if it would work for humans as well. But the research is up-and-coming. “We can now generate insulin-producing cells that look and act a lot like the pancreatic beta cells you and I have in our bodies. This is a critical step towards our goal of creating cells that could be transplanted into patients with diabetes,” He said.
“We’re finally able to move forward on several different fronts that were previously closed to us,” he added. “The possibilities seem endless.”
Basic research keeps elucidating new aspects of beta cells; there seem to be several subtypes, so the gold standard for duplicating the cells is not entirely clear. Today, however, there is “a handful of groups in the world that can generate a cell that looks like a beta cell,” says Hebrok, who currently acts as scientific advisor to Semma and Sigilon, and has previously advised ViaCyte. “Certainly, companies have convinced themselves that what they have achieved is good enough to go into patients.”
The stem cell reprogramming methods that the three companies use to prompt cell differentiation create a mixture of islet cells. Beta cells sit in pancreatic islets of Langerhans alongside other types of endocrine cells. Alpha cells, for example, churn out glucagon, a hormone that stimulates the conversion of glycogen into glucose in the liver and raises blood sugar. Although the companies agree on the positive potential of islet cell mixtures, they take different approaches to developing and differentiating their cells. Semma, which was launched in 2014 to commercialize the Harvard group’s work and counts Novartis among its backers, describes its cells as fully mature, meaning that they are wholly differentiated into beta or other cells before transplantation. “Our cells are virtually indistinguishable from the ones you would isolate from donors,” says Semma chief executive officer BastianoSanna
To get around the donor problem, researchers, including the team at UCSF has been working on nudging stem cells into becoming fully-functional pancreatic beta cells for the last few years. Still, there have been some issues in getting them all the way there.
“The cells we and others were producing were getting stuck at an immature stage where they weren’t able to respond adequately to blood glucose and secrete insulin properly,” Hebrok said.
“It has been a major bottleneck for the field.”
“We’re finally able to move forward on a number of different fronts that were previously closed to us,” Hebrok added. “The possibilities seem endless.”
Regardless of starting cell type, the companies say they are ready to churn out their cells in large numbers. Semma, for example, can make more islet cells in a month than can be isolated from donors in a year in the United States, Sanna says, and the company’s “pristine” cells should perform better than donor islets, which are battered by the aggressive techniques required for their isolation.
As these products, some of which have already entered clinical trials, move toward commercialization, regulatory agencies such as the US Food and Drug Administration (FDA) and the European Medicines Agency have expressed concern about the plasticity of the reprogrammed cells. All three firms subject their cells to rigorous safety testing to ensure that they don’t turn tumorigenic. Before successful trials, companies won’t know the dose of beta cells required for a functional cure, or how long such “cures” will last before needing to be boosted. There’ll be commercial challenges, too: while the companies are investing heavily to develop suitable industrial processes, all acknowledge that no organization has yet manufactured cell therapies in commercial volumes.
Nevertheless, there’s growing confidence throughout the field that these problems will be solved, and soon. “We have the islet cells now,” says Alice Tomei, a biomedical engineer at the University of Miami who directs DRI’s Islet Immuno-engineering Laboratory.
“These stem cell companies are working hard to try to get FDA clearance on the cells.”
Protecting stem cell therapies from the immune system
Whatever the type of cell being used, another major challenge is delivering cells to the patient in a package that guards against immune attack while keeping cells fully functional. Companies are pursuing two main strategies:
- Microencapsulation, where cells are immobilized individually or as small clusters, in tiny blobs of a biocompatible gel.
- Macroencapsulation, in which greater numbers of cells are put into a much larger, implantable device.
ViaCyte, which recently partnered with Johnson & Johnson, launched its first clinical trial in 2014. The trial involved a micro-encapsulation approach that packaged up the company’s partially differentiated, ESC-derived cells into a flat device called the PEC-Encapsulation. About the size of a Band-Aid, the device is implanted under the skin, where the body forms blood vessels around it. “It has a semipermeable membrane that allows the free flow of oxygen, nutrients, and glucose,” says ViaCyte’s chief executive officer, Paul Laikind. “And even proteins like insulin and glucagon can move back and forth across that membrane, but cells cannot.”
The trial showed that the device was safe, well-tolerated, and protected from the adaptive immune system—and that some cells differentiated into working islet cells. But most cells didn’t engraft effectively because a “foreign body response,” a variant of wound healing, clogged the PEC-Encap’s membrane and prevented vascularization. ViaCyte stopped the trial and partnered with W. L. Gore & Associates, the maker of Gore-Tex, to engineer a new membrane. “With this new membrane,” says Laikind, “we’re not eliminating that foreign body response, but we’re overcoming it in such a way that allows vascularization to take place.” The company expects to resume the trial in the second half of this year, provided it receives the green light from the FDA.
Semma is also developing macro¬-encapsulation methods, including a very thin device that in prototype form is about the size of a silver dollar coin. The device is “deceptively simple, but it allows us to put [in] a fully curative dose of islets,” Sanna says.
Semma is also investigating microencapsulation alternatives. At the same time, the company is advancing toward clinical trials using established transplantation techniques to administer donated cadaver cells to high-risk patients who find it particularly difficult to control their blood glucose levels. These cells are infused via the portal vein into the liver, and patients take immunosuppressive drugs to prevent rejection.
Sigilon is working on its microencapsulation technology. Launched in 2016 on the back of work by the labs of Robert Langer and Daniel Anderson at MIT, the company has created 1.5-millimeter gel-based spheres that can hold between 5,000 and 30,000 cells (Nat Med, 22:306–11, 2016). Each sphere is like a balloon, with the outside chemically modified to provide immune-protection, says Sigilon chief executive officer Rogerio Vivaldi. “The inside of the balloon is full of a gel that creates almost a kind of a matrix net where the cells reside.”
In 2018, shortly after partnering with Eli Lilly, Sigilon and collaborators published research showing that islet cells that were encapsulated in gel spheres and transplanted into macaques remained functional for four months. The company has not disclosed a time frame for a type 1 diabetes trial “but we’re moving pretty quickly,” says chief scientific officer David Moller.
Conclusion
To conclude, all three firms hope to extend their work to treat some of the 400 million people worldwide with type 2 diabetes, many of them eventually benefit from insulin injections. The recent endorsements from big Pharmaceutical underline the real progress in beta-cell transplants, says Aaron Kowalski, a molecular geneticist and chief executive officer at JDRF, a foundation based in New York that has funded research at ViaCyte and academic labs whose work has been tapped by Semma and Sigilon. “These companies all realize that if they don’t do it, somebody else will. It’s hard to predict exactly when, but somebody is going to make this work.”
- Published in Corporate News / Blog
Conventional and novel stem cell based therapies for androgenic alopecia
Dodanim Talavera-Adame,1 Daniella Newman,2 Nathan Newman1
1American Advanced Medical Corp. (Private Practice), Beverly Hills, CA,
2Western University of Health Sciences, Pomona, CA, USA
Abstract
The prevalence of androgenic alopecia (AGA) increases with age and it affects both men and women. Patients diagnosed with AGA may experience decreased quality of life, depression, and feel self-conscious. There are a variety of therapeutic options ranging from prescription drugs to non-prescription medications. Currently, AGA involves an annual global market revenue of US$4 billion and a growth rate of 1.8%, indicating a growing consumer market. Although natural and synthetic ingredients can promote hair growth and, therefore, be useful to treat AGA, some of them have important adverse effects and unknown mechanisms of action that limit their use and benefits. Biologic factors that include signaling from stem cells, dermal papilla cells, and platelet-rich plasma are some of the current therapeutic agents being studied for hair restoration with milder side effects. However, most of the mechanisms exerted by these factors in hair restoration are still being researched. In this review, we analyze the therapeutic agents that have been used for AGA and emphasize the potential of new therapies based on advances in stem cell technologies and regenerative medicine.
Introduction
The prevalence of androgenic alopecia (AGA) increases with age, and is estimated to affect about 80% of Caucasian men.1 Female AGA, also known as female pattern hair loss, affects 32% of women in the ninth decade of life.2 The consumer market for products that promote hair growth has been increasing dramatically.3 These products promote hair regeneration based on the knowledge about the hair follicle (HF) cycle.4,5 However, in most cases, the mechanisms of action of these products are not well characterized and the results are variable or with undesirable side effects.6 At present, only two treatments for AGA have been approved by the US Food and Drug Administration (FDA): Minoxidil and Finasteride.7–10Although these medications have proved to be effective in some cases, their use is limited by their side effects.11,12 With the emergence of stem cells (SCs), many mechanisms that lead to tissue regeneration have been discovered.13 Hair regeneration has become one of the targets for SC technologies to restore the hair in AGA.14 Several SC factors such as peptides exert essential signals to promote hair regrowth.15,16 Some of these signals stimulate differentiation of SCs to keratinocytes which are important for HF growth.17 Other signals can stimulate dermal papilla cells (DPCs) that promote SC proliferation in the HF.18,19 In this review, we describe HF characteristics and discuss different therapies used currently for AGA and possible novel agents for hair regeneration. These therapies include FDA-approved medications, non-prescription physical or chemical agents, natural ingredients, small molecules, biologic factors, and signals derived from SCs.
HF and SC niche
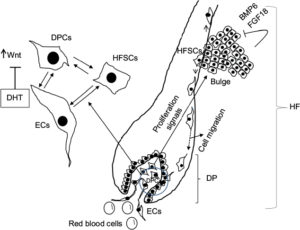
The HF undergoes biologic changes from an actively growing stage (anagen) to a quiescent stage (telogen) with an intermediate remodeling stage (catagen).4 HFSCs are located in the bulge region of the follicle and they interact with mesenchymal SCs (MSCs) located in the dermal papilla (DP).18 These signal exchanges promote activation of some cellular pathways that are essential for DPC growth, function, and survival, such as the activation of Wnt signaling pathway.19–21 Other signals, such as those from endothelial cells (ECs) located at the DP, are also essential for HF maintenance.22 EC dysfunction that impairs adequate blood supply may limits or inhibits hair growth.22 For instance, Minoxidil, a synthetic agent, is able to promote hair growth by increasing blood flow and the production of prostaglandin E2 (PGE2).7 It has been shown that proteins that belong to the transforming growth factor (TGF) superfamily, such as bone morphogenetic proteins (BMPs), also exert signals to maintain the capacity of DPCs to induce HF growing in vivo and in vitro.23 These BMPs may be released by several cells that compose the follicle, including ECs.24–26 ECs may provide signals for BMP receptor activation in DPCs similar to those signals that promote survival of MSCs in human embryoid bodies composed of multipotent cells.24,25 DPCs have been derived from pluripotent SCs in an attempt to study their potential for hair regeneration in vitro and in vivo.27 Together, dermal blood vessels and DPCs orchestrate a suitable microenvironment for the growth and survival of HFSCs.28,29 Interestingly, the expression of Forkhead box C1 regulates the quiescence of HFSCs located in the bulge region (Figure 1).30 HFSCs are quiescent during mid-anagen and maintain this stage until the next hair cycle.29,30 However, during early anagen stage, these cells undergo a short proliferative phase in which they self-renew and produce new hair.30 Therefore, the bulge region constitutes a SC niche that makes multiple signals toward quiescence or proliferation stages.30–34 It is known that fibroblasts and adipocyte signals are able to inhibit the proliferation of HFSCs.34 Additionally, BMP6 and fibroblast growth factor 18 (FGF18) from bulge cells exert inhibitory effects on HFSC proliferation.34 Dihydrotestosterone (DHT) also inhibits HF growth.35 Agents that reduce DHT, such as Finasteride, promote hair regrowth by inhibiting Type II 5a-reductase.8,14,36 In contrast to these inhibitory effects, DPCs located at the base of the HF provide activation signals (Figure 1).18,34 The crosstalk between DPCs and HFSCs leads to inhibition of inhibitory effects with the resultant cell proliferation toward hair regeneration (anagen).30,31,37 With the self-renewal of HFSCs, the outer root sheath (ORS) forms, and signals from DPCs to the bulge cells diminish in a way that the bulge cells start again with their quiescent stage.4,34As mentioned earlier, Forkhead box C1 transcription factor has an important role in maintaining the threshold for HFSC activation.30 The knockdown of these factors in bulge cells reduces the cells’ threshold for proliferation, and the anagen cycle starts more frequently due to promotion of HFSC proliferation in shorter periods of time.30
Laser therapy
Light amplification by stimulated emission of radiation (LASER) generates electromagnetic radiation which is uniform in polarization, phase, and wavelength.45 Low-level laser therapy (LLLT), also called “cold laser” therapy, since it utilizes lower power densities than those needed to produce heating of tissue. Transdermal LLLT has been used for therapeutic purposes via photobiomodulation.46,47 Several clinical conditions, such as rheumatoid arthritis, mucositis, pain, and other inflammatory diseases, have been treated with these laser devices.48–50 LLLT promotes cell proliferation by stimulating cellular production of adenosine triphosphate and creating a shift in overall cell redox potential toward greater intracellular oxidation.51 The redox state of the cell regulates activation of signaling pathways that ultimately promotes high transcription factor activity and gene expression of factors associated with the cell cycle.52 Physical agents such as lasers have been also used to prevent hair loss in a wavelength range in the red and near infrared (600–1,070 nm).5,47,51,53 Laser therapy emits light that penetrates the scalp and promotes hair growth by increasing the blood flow.54 This increase gives rise to EC proliferation and migration due to upregulation of vascular endothelial growth factor (VEGF) and endothelial nitric oxide synthase.55,56 In addition, the laser energy itself stimulates metabolism in catagen or telogen follicles, resulting in the production of anagen hair.53,54A specific effect of LLLT has been demonstrated to promote proliferation of HFSCs, forcing the hair to start the anagen phase.57
Biologic agents that promote hair growth and their mechanisms of action
SC signaling
Recently, it has been found that SCs release factors that can promote hair growth.16 These factors and their mechanisms of action have been summarized in Table 3. These factors, known as “secretomes”, are able to promote skin regeneration, wound healing, and immunologic modulation, among other effects.58,59 Some of these factors, such as epidermal growth factor (EGF), basic fibroblast growth factor, hepatocyte growth factor (HGF) and HGF activator, VEGF, insulin-like growth factor (IGF), TGF-ß, and platelet-derived growth factor (PDGF), are able to provide signals that promote hair growth.15,60–64 As mentioned before, DPCs provide signals to HFSCs located in the bulge that proliferate and migrate either to the DP or to the epidermis to repopulate the basal layer (Figure 1).32,65 Enhancement in growth factor expression (except for EGF) has been reported when the adipose SCs are cultured in hypoxic conditions.15 Also, SCs increase their self-renewal capacity under these conditions.66–68 Low oxygen concentrations (1%–5%) increase the level of expression of SC factors that include VEGF, basic fibroblast growth factor, IGF binding protein 1 (IGFBP-1), IGF binding protein 2 (IGFBP-2), macrophage colony-stimulating factor (M-CSF), M-CSF receptor (M-CSFR), and PDGF receptor ß (PDGFR-ß).15,69,70 While these groups of factors promote HF growth in intact skin, another group of factors, such as M-CSF, M-CSFR, and interleukin-6, are involved in wound-induced hair neogenesis.71 HGF and HGF activator stimulate DPCs to promote proliferation of epithelial follicular cells.61 Epidermal growth factor promotes cellular migration via the activation of Wnt/ß-catenin signaling.60 VEGF promotes hair growth and increases the follicle size mainly by perifollicular angiogenesis.72 Blocking VEGF activity by neutralizing antibodies reduced the size and growth of the HF.72 PDGF and its receptor (PDGFR-a) are essential for follicular development by promoting upregulation of genes involved in HF differentiation and regulating the anagen phase in HFs.64,73 They are also expressed in neonatal skin cells that surround the HF.73 Monoclonal antibodies to PDGFR-a (APA5) produced failure in hair germ induction, supporting that PDGFR-a and its ligand have an essential role in hair differentiation and development.73 IGF-1 promotes proliferation, survival, and migration of HF cells.69,74 In addition, IGF binding proteins (IGFBPs) also promote hair growth and hair cell survival by regulating IGF-1 effects and its interaction with extracellular matrix proteins in the HF.70 Higher levels of IGF-1 and IGFBPs in beard DPCs suggest that IGF-1 levels are associated with androgens.74 Furthermore, DPCs from non-balding scalps showed significantly higher levels of IGF-1 and IGFBP-6, in contrast to DPCs from balding scalps.74
Table 3
Stem cell factors and small molecules that promote hair growth and their mechanisms of action
| Factor | Mechanism of action |
|---|---|
| HGF and HGF activator61 | Factor secreted by DPC that promotes proliferation of epithelial follicular cells |
| EGF60 | Promotes growth and migration of follicle ORS cells by activation of Wnt/ß-catenin signaling |
| bFGF62 | Promotes the development of hair follicle |
| IL-693 | Involved in WIHN through STAT3 activation |
| VEGF72 | Promotes perifollicular angiogenesis |
| TGF-ß63 | Stimulates the signaling pathways that regulate hair cycle |
| IGF-169 | Promotes proliferation, survival, and migration of hair follicle cells |
| IGFBP-1 to -670 | Regulates IGF-1 effects and its interaction with extracellular matrix proteins at the hair follicle level |
| BMP23 | Maintains DPC phenotype which is crucial for stimulation of hair follicle stem cell |
| BMPR1a23 | Maintains the proper identity of DPCs that is essential for specific DPC function |
| M-CSF71 | Involved in wound-induced hair regrowth |
| M-CSFR71 | Involved in wound-induced hair regrowth |
| PDGF and PDGFR-ß/-a64 | Upregulates the genes involved in hair follicle differentiation. Induction and regulation of anagen phase. PDGF and its receptors are essential for follicular development |
| Wnt3a97 | Involved in hair follicle development through ß-catenin signaling |
| PGE279,80 | Stimulates anagen phase in hair follicles |
| PGF2a and analogs79,80 | Promotes transition from telogen to anagen phase of the hair cycle |
| BIO98 | GSK-3 inhibitor |
| PGE2 or inhibition of PGD2 or PGD2 receptor D2/GPR4477 | Promotes follicle regeneration |
| Iron and l-lysine95 | Under investigation |
Abbreviations: bFGF, basic fibroblast growth factor; BIO, (2’Z,3’E)-6-bromoindirubin-3′-oxime; BMP, bone morphogenetic protein; DPCs, dermal papilla cells; EGF, epidermal growth factor; GSK-3, glycogen synthase kinase-3; HGF, hepatocyte growth factor; IGF-1, insulin-like growth factor 1; IGFBP-1, insulin-like growth factor-binding protein 1; IL-6, interleukin-6; M-CSF, microphage colony-stimulating factor; M-CSFR, microphage colony-stimulating factor receptor; ORS, outer root sheath; PDGF, platelet-derived growth factor; PDGFR-a, platelet-derived growth factor receptor alpha; PDGFR-ß, platelet-derived growth factor receptor beta; PGD2, prostaglandin D2; PGE2, prostaglandin E2; TGF-ß1, transforming growth factor ß1; VEGF, vascular endothelial growth factor; WIHN, wound-induced hair neogenesis; Wnt3a, wingless-type MMTV integration site family, member 3A.
Small molecules
Small molecules with low molecular weight (<900 Da) and the size of 10-9 m are organic compounds that are able to regulate some biologic processes.75 Some small molecules have been tested for their role in hair growth.76 Synthetic, non-peptidyl small molecules that act as agonists of the hedgehog pathway have the ability to promote follicular cycling in adult mouse skin.76 PGE2 and prostaglandin D2 (PGD2) have also been associated with the hair cycle (Table 3).77 PGD2 is elevated in the scalp of balding men and inhibits hair lengthening via GPR44 receptor.78 Also, it is known that PGE2 and PGF2a promote hair growth, while PGD2 inhibits this process.77,79 Prostaglandin analogs of PGF2a have been used originally to decrease ocular pressure in glaucoma with parallel effects in the growth of eyelashes, which suggests a specific effect in HF activation.80 PGD2 receptors are located in the upper and lower ORS region and in the DP, suggesting that these prostaglandins play an important role in hair cycle.81 Molecules such as quercetin are able to inhibit PGD2 and, in this way, promote hair growth.82–84 Antagonists of PGD2 receptor (formally named chemoattractant receptor-homologous expressed in Th2 cells) such as setipiprant have been used to treat allergic diseases such as asthma, but they also have beneficial effects in AGA.85–87 Another small molecule l-ascorbic acid 2-phosphate promotes proliferation of ORS keratinocytes through the secretion of IGF-1 from DPCs via phosphatidylinositol 3-kinase.88 Recently, it has been described that small-molecule inhibitors of Janus kinase–signal transducer and activator of transcription (JAK-STAT) pathway promote hair regrowth in humans.89 Janus kinase inhibitors are currently approved by the FDA for the treatment of some specific diseases such as psoriasis and other autoimmune-mediated diseases.90–94 Also, another group of small molecules such as iron and the amino acid l-Lysine are essential for hair growth (Table 3).95
Cellular therapy
The multipotent SCs in the bulge region of the HF receive signals from DPCs in order to proliferate and survive.27,28,65,84,96 It has been shown that Wnt/ß-catenin signaling is essential for the growth and maintenance of DPCs.19,97 These cells can be isolated and cultured in vitro with media supplemented with 10% fetal bovine serum and FGF-2.37,98 However, they lose versican expression that correlates with decrease in follicle-inducing activity in culture.98 Versican is the most abundant component of HF extracellular matrix.99 Inhibition of glycogen synthase kinase-3 by (2’Z,3’E)-6-bromoindirubin-3′-oxime (BIO) promotes hair growth in mouse vibrissa follicles in culture by activation of Wnt signaling.98 Therefore, the increase of Wnt signaling in DPCs apparently is one of the main factors that promote hair growth.19 DPCs have been also generated from human embryonic SCs that induced HF formation after murine transplantation.27
Platelet-rich plasma
Platelets are anucleate cells generated by fragmentation of megakaryocytes in the bone marrow.100 These cells are actively involved in the hemostatic process after releasing biologically active molecules (cytokines).100–102 Because of the platelets’ higher capacity to produce and release these factors, autologous platelet-rich plasma (PRP) has been used to treat chronic wounds.103 Therefore, PRP can be used as autologous therapy for regenerative purposes, for example, chondrogenic differentiation, wound healing, fat grafting, AGA, alopecia areata, facial scars, and dermal volume augmentation.101,104–108 PRP contains human platelets in a small volume that is five to seven times higher than in normal blood and it has been proven to be beneficial to treat AGA.10,105,109–111 The factors released by these platelets after their activation, such as PDGFs (PDGFaa, PDGFbb, PDGFab), TGF-ß1, TGF-ß2, EGF, VEGF, and FGF, promote proliferation of DPCs and, therefore, may be beneficial for AGA treatment.109,112–114 Clinical experiments indicate that patients with AGA treated with autologous PRP show improved hair count and thickness.109
In search of novel therapies
In this paper, we reviewed and discussed the use of therapeutic agents for hair regeneration and the knowledge to promote the development of new therapies for AGA based on the advances in regenerative medicine. The HF is a complex structure that grows when adequate signaling is provided to the HFSCs. These cells are located in the follicle bulge and receive signals from MSCs located in the dermis that are called DPCs. The secretory phenotype of DPCs is determined by local and circulatory signals or hormones. Recent discoveries have demonstrated that SCs in culture are able to activate DPCs and HFSCs and, in this way, promote hair growth. The study of these cellular signals can provide the necessary knowledge for developing more effective therapeutic agents for the treatment of AGA with minimal side effects. Therefore, advancements in the field of regenerative medicine may generate novel therapeutic alternatives. However, further research and clinical studies are needed to evaluate their efficacy.
Disclosure
The authors report no conflicts of interest in this work.
References
|
Blumeyer A, Tosti A, Messenger A, et al; European Dermatology Forum (EDF). Evidence-based (S3) guideline for the treatment of androgenetic alopecia in women and in men. J Dtsch Dermatol Ges. 2011;9(Suppl 6):S1–S57. |
||
|
Ramos PM, Miot HA. Female pattern hair loss : a clinical and pathophysiological review. An Bras Dermatol. 2015;90(4):529–543. |
||
|
Otberg N, Finner AM, Shapiro J. Androgenetic alopecia. Endocrinolo Metab Clin North Am. 2007;36(2):379–398. |
||
|
Plikus MV, Chuong CM. Complex hair cycle domain patterns and regenerative hair waves in living rodents. J Invest Dermatol. 2008;128(5):1071–1080. |
||
|
McElwee KJ, Shapiro JS. Promising therapies for treating and/or preventing androgenic alopecia. Skin Therapy Lett. 2012;17(6):1–4. |
||
|
Perez-Mora N, Velasco C, Bermüdez F. Oral finasteride presents with sexual-unrelated withdrawal in long-term treated androgenic alopecia in men. Skinmed. 2015;13(3):179–183. |
||
|
Gupta AK, Charrette A. Topical minoxidil: systematic review and meta-analysis of its efficacy in androgenetic alopecia. Skinmed. 2015;13(3):185–189. |
||
|
Shapiro J, Kaufman KD. Use of finasteride in the treatment of men with androgenetic alopecia (male pattern hair loss). J Investig Dermatol Symp Proc. 2003;8(1):20–23. |
||
|
Rossi A, Anzalone A, Fortuna MC, et al. Multi-therapies in androgenetic alopecia: review and clinical experiences. Dermatol Ther. 2016;29(6):424–432. |
||
|
Varothai S, Bergfeld WF. Androgenetic alopecia: an evidence-based treatment update. Am J Clin Dermatol. 2014;15(3):217–230. |
||
|
Rossi A, Cantisani C, Melis L, Iorio A, Scali E, Calvieri S. Minoxidil use in dermatology, side effects and recent patents. Recent Pat Inflamm Allergy Drug Discov. 2012;6(2):130–136. |
||
|
Motofei IG, Rowland DL, Georgescu SR, Mircea T, Baleanu BC, Paunica S. Are hand preference and sexual orientation possible predicting factors for finasteride adverse effects in male androgenic alopecia? Exp Dermatol. 2016;25(7):557–558. |
||
|
Reijo Pera RA, Gleeson JG. Stems cells and regeneration: special review issue. Hum Mol Gen. 2008;17(R1):R1–R2. |
||
|
Jain R, De-Eknamkul W. Potential targets in the discovery of new hair growth promoters for androgenic alopecia. Expert Opin Ther Targets. 2014;18(7):787–806. |
||
|
Park BS, Kim WS, Choi JS, et al. Hair growth stimulated by conditioned medium of adipose-derived stem cells is enhanced by hypoxia : evidence of increased growth factor secretion. Biomed Res. 2010;31(1):27–34. |
||
|
Fukuoka H, Suga H. Hair regeneration treatment using adipose-derived stem cell conditioned medium :follow-up with trichograms. Eplasty. 2015;15:65–72. |
||
|
Du Y, Roh DS, Funderburgh ML, et al. Adipose-derived stem cells differentiate to keratocytes in vitro. Mol Vis. 2010;16:2680–2689. |
||
|
Zhang P, Kling RE, Ravuri SK, et al. A review of adipocyte lineage cells and dermal papilla cells in hair follicle regeneration. J Tissue Eng. 2014;5:2041731414556850. |
||
|
Tsai SY, Sennett R, Rezza A, et al. Wnt/ß-catenin signaling in dermal condensates is required for hair follicle formation. Dev Biol. 2014;385(2):179–188. |
||
|
Choi YS, Zhang Y, Xu M, et al. Distinct functions for Wnt/ß-catenin in hair follicle stem cell proliferation and survival and interfollicular epidermal homeostasis. Cell Stem Cell. 2013;13(6):720–733. |
||
|
Rao TP, Kuhl M. An updated overview on Wnt signaling pathways a prelude for more. Circ Res. 2010;106(12):1798–1806. |
||
|
Yano K, Brown LF, Detmar M. Control of hair growth and follicle size by VEGF-mediated angiogenesis. J Clin Invest. 2001;107(4):409–417. |
||
|
Rendl M, Polak L, Fuchs E. BMP signaling in dermal papilla cells is required for their hair follicle-inductive properties. Genes Dev. 2008;22(4):543–557. |
||
|
Talavera-Adame D, Gupta A, Kurtovic S, Chaiboonma KL, Arumugaswami V, Dafoe DC. Bone morphogenetic protein-2/-4 upregulation promoted by endothelial cells in coculture enhances mouse embryoid body differentiation. Stem Cells Dev. 2013;22(24):3252–3260. |
||
|
Talavera-Adame D, Wu G, He Y, et al. Endothelial Cells in Co-culture Enhance Embryonic Stem Cell Differentiation to Pancreatic Progenitors and Insulin-Producing Cells through BMP Signaling. Stem Cell Rev. 2011;7(3):532–543. |
||
|
Talavera-Adame D, Ng TT, Gupta A, Kurtovic S, Wu GD, Dafoe DC. Characterization of microvascular endothelial cells isolated from the dermis of adult mouse tails. Microvascular Res. 2011;82(2):97–104. |
||
|
Gnedeva K, Vorotelyak E, Cimadamore F, et al. Derivation of hair-inducing cell from human pluripotent stem cells. PLoS One. 2015;10(1):1–14. |
||
|
Lavker RM, Sun T, Oshima H, et al. Hair follicle stem cells. J Investig Dermatol Symp Proc. 2003;8(1):28–38. |
||
|
Bernard B. Advances in understanding hair growth. F1000Res. 2016;5:1–8. |
||
|
Lay K, Kume T, Fuchs E. FOXC1 maintains the hair follicle stem cell niche and governs stem cell quiescence to preserve long-term tissue-regenerating potential. Proc Natl Acad Sci U S A. 2016;113(1): E1506–E1515. |
||
|
Leirós GJ, Attorresi AI, Balañá ME. Hair follicle stem cell differentiation is inhibited through cross-talk between Wnt/ß-catenin and androgen signalling in dermal papilla cells from patients with androgenetic alopecia. Br J Dermatol. 2012;166(5):1035–1042. |
||
|
Chacon-Martinez CA, Klose M, Niemann C, Glauche I, Wickström SA. Hair follicle stem cells= cultures reveal self-organizing plasticity of stem cells and their progeny. EMBO J. 2017;36(2):151–164. |
||
|
Soler AP, Gilliard G, Megosh LC, O’Brien TG. Modulation of murine hair follicle function by alterations in ornithine decarboxylase activity. J Invest Dermatol. 1996;106(5):1108–1113. |
||
|
Hsu YC, Pasolli HA, Fuchs E. Dynamics between stem cells, nich and progeny in the hair follicle. Cell. 2011;144(1):92–105. |
||
|
Kang JI, Kim SC, Kim MK, et al. Effects of dihydrotestosterone on rat dermal papilla cells in vitro. Eur J Pharmacol. 2015;757:74–83. |
||
|
Yamana K, Labrie F, Luu-The V. Human type 3 5a-reductase is expressed in peripheral tissues at higher levels than types 1 and 2 and its activity is potently inhibited by finasteride and dutasteride. Horm Mol Biol Clin Investig. 2010;2(3):293–299. |
||
|
Osada A, Iwabuchi T, Kishimoto J, Hamazaki TS, Okochi H. Long-term culture of mouse vibrissal dermal papilla cells and de novo hair follicle induction. Tissue Eng. 2007;13(5):975–982. |
||
|
Choi GS, Kim JH, Oh SY, et al. Safety and tolerability of the dual 5-alpha reductase inhibitor dutasteride in the treatment of androgenetic alopecia. Ann Dermatol. 2016;28(4):444–450. |
||
|
Carson C 3rd, Rittmaster R. The role of dihydrotestosterone in benign prostatic hyperplasia. Urology. 2003;61(4 Suppl 1):2–7. |
||
|
Kamimura A, Takahashi T. Procyanidin B-2, extracted from apples, promotes hair growth: a laboratory study. Br J Dermatol. 2002;146(1):41–51. |
||
|
Kamimura A, Takahashi T. Procyanidin B-3, isolated from barley and identified as a hair-growth stimulant, has the potential to counteract inhibitory regulation by TGF-beta1. Exp Dermatol. 2002;11(6):532–541. |
||
|
Ulbricht C, Basch E, Bent S, et al. Evidence-based systematic review of saw palmetto by the Natural Standard Research Collaboration. J Soc Integr Oncol. 2006;4(4):170–186. |
||
|
Rondanelli M, Perna S, Peroni G, Guido D. A bibliometric study of scientific literature in Scopus on botanicals for treatment of androgenetic alopecia. J Cosmet Dermatol. 2015;15(2):120–130. |
||
|
Blume-Peytavi U, Kunte C, Krisp A, Garcia Bartels N, Ellwanger U, Hoffmann R. [Comparison of the efficacy and safety of topical minoxidil and topical alfatradiol in the treatment of androgenetic alopecia in women.] J Dtsch Dermatol Ges. 2007;5(5):391–395. German. |
||
|
Farivar S, Malekshahabi T, Shiari R. Biological effects of low level laser therapy. J Lasers Med Sci. 2014;5(2):58–62. |
||
|
Maiman TH. Biomedical lasers evolve toward clinical applications. Hosp Manage. 1966;101(4):39–41. |
||
|
Cung H, Dai T, Sharma SK, et al. The nuts and bolts of low-level laser (light) therapy. Ann Biomed Eng. 2013;40(2):516–533. |
||
|
Bjordal JM, Couppé C, Chow RT, Tunér J, Ljunggren EA. A systematic review of low level laser therapy with location-specific doses for pain from chronic joint disorders. The Aust J Physiother. 2003;49(2):107–116. |
||
|
Brosseau L, Welch V, Wells G, et al. Low level laser therapy (classes I, II and III) in the treatment of rheumatoid arthritis. Cochrane Database Syst Rev. 2005;19(2):CD002049. |
||
|
Cauwels RGEC, Martens LC. Low level laser therapy in oral mucositis: a pilot study. Eur Arch Paediatr Dent. 2011;12(2):118–123. |
||
|
Schindl A, Schindl M, Pernerstorfer-Schön H, Schindl L. Low-intensity laser therapy: a review. J Investig Med. 2000;48(5):312–326. |
||
|
Liu H, Colavitti R, Rovira II, Finkel T. Redox-dependent transcriptional regulation. Cir Res. 2005;97(10):967–974. |
||
|
Gupta AK, Lyons DCA, Abramovits W. Low-level laser/light therapy for androgenetic alopecia. Skinmed. 2014;12(3):145–147. |
||
|
Jimenez JJ, Wikramanayake TC, Bergfeld W, et al. Efficacy and safety of a low-level laser device in the treatment of male and female pattern hair loss: a multicenter, randomized, sham device-controlled, double-blind study. Am J Clin Dermat. 2014;15(2):115–127. |
||
|
Chen CH, Hung HS, Hsu SH. Low-energy laser irradiation increases endothelial cell proliferation, migration, and eNOS gene expression possibly via PI3K signal pathway. Lasers Surg Med. 2008;40(1):46–54. |
||
|
Tuby H, Maltz L, Oron U. Modulations of VEGF and iNOS in the rat heart by low level laser therapy are associated with cardioprotection and enhanced angiogenesis. Lasers Surg Med. 2006;38(7):682–688. |
||
|
Avci P, Gupta GK, Clark J, Wikonkal N, Hamblin MR. Low-level laser (light) therapy (LLLT) for treatment of hair loss. Laser Surg Med. 2015;46(2):144–151. |
||
|
Rodrigues C, de Assis AM, Moura DJ, et al. New therapy of skin repair combining adipose-derived mesenchymal stem cells with sodium carboxymethylcellulose scaffold in a pre-clinical rat model. PloS One. 2014;9(5):e96241. |
||
|
Gimble JM, Katz AJ, Bunnell BA. Adipose-derived stem cells for regenerative medicine. Cir Res. 2007;100(9):1249–1260. |
||
|
Zhang H, Nan W, Wang S, et al. Epidermal growth factor promotes proliferation and migration of follicular outer root sheath cells via Wnt/ß-catenin signaling. Cell Physiol Biochem. 2016;39(1):360–370. |
||
|
Lee YR, Yamazaki M, Mitsui S, Tsuboi R, Ogawa H. Hepatocyte growth factor (HGF) activator expressed in hair follicles is involved in in vitro HGF-dependent hair follicle elongation. J Dermatol Sci. 2001;25(2):156–163. |
||
|
du Cros DL. Fibroblast growth factor influences the development and cycling of murine hair follicles. Dev Biol. 1993;156(2):444–453. |
||
|
Niimori D, Kawano R, Felemban A, et al. Tsukushi controls the hair cycle by regulating TGF-ß1 signaling. Dev Biol. 2012;372(1):81–87. |
||
|
Tomita Y, Akiyama M, Shimizu H. PDGF isoforms induce and maintain anagen phase of murine hair follicles. J Dermatol Sci. 2006;43(2):105–115. |
||
|
Ohyama M, Terunuma A, Tock CL, et al. Characterization and isolation of stem cell – enriched human hair follicle bulge cells. J Clin Invest. 2006;116(1):249–260. |
||
|
Grayson WL, Zhao F, Bunnell B, Ma T. Hypoxia enhances proliferation and tissue formation of human mesenchymal stem cells. Biochem Biophys Res Commun. 2007;358(3):948–953. |
||
|
Potier E, Ferreira E, Andriamanalijaona R, et al. Hypoxia affects mesenchymal stromal cell osteogenic differentiation and angiogenic factor expression. Bone. 2007;40(4):1078–1087. |
||
|
Ren H, Cao Y, Zhao Q, et al. Proliferation and differentiation of bone marrow stromal cells under hypoxic conditions. Biochem Biophys Res Commun. 2006;347(1):12–21. |
||
|
Su HY, Hickford JG, Bickerstaffe R, Palmer BR. Insulin-like growth factor 1 and hair growth. Dermatol Online J. 1999;5(2):1. |
||
|
Batch JA, Mercuri FA, Werther GA. Identification and localization of insulin-like growth factor-binding protein (IGFBP) messenger RNAs in human hair follicle dermal papilla. J Invest Dermatol. 1996;106(3):471–475. |
||
|
Osaka N, Takahashi T, Murakami S, et al. ASK1-dependent recruitment and activation of macrophages induce hair growth in skin wounds. J Cell Biol. 2007;176(7):903–909. |
||
|
Yano K, Brown LF, Detmar M. Control of hair growth and follicle size by VEGF-mediated angiogenesis. J Clin Invest. 2001;107(4):409–417. |
||
|
Takakura N, Yoshida H, Kunisada T, Nishikawa S, Nishikawa SI. Involvement of platelet-derived growth factor receptor-a in hair canal formation. J Invest Dermatol. 1996;107(5):770–777. |
||
|
Panchaprateep R, Asawanonda P. Insulin-like growth factor-1: roles in androgenetic alopecia. Exp Dermatol. 2014;23(3):216–218. |
||
|
Castoreno AB, Eggert US. Small molecule probes of cellular pathways and networks. ACS Chem Biol. 2011;6(1):86–94. |
||
|
Paladini RD, Saleh J, Qian C, Xu GX, Rubin LL. Modulation of hair growth with small molecule agonists of the hedgehog signaling pathway. J Iinvest Dermatol. 2005;125(4):638–646. |
||
|
Nieves A, Garza LA. Does prostaglandin D2 hold the cure to male pattern baldness? Exp Dematol. 2014;(29):224–227. |
||
|
Nelson AM, Loy DE, Lawson JA, Katseff AS, Fitzgerald GA, Garza LA. Prostaglandin D2 inhibits wound-induced hair follicle neogenesis through the receptor Gpr44. J Invest Dermatol. 2013;133(4):881–889. |
||
|
Sasaki S, Hozumi Y, Kondo S. Influence of prostaglandin F2alpha and its analogues on hair regrowth and follicular melanogenesis in a murine model. Exp Dermatol. 2005;14(5):323–328. |
||
|
Choi YM, Diehl J, Levins PC. Promising alternative clinical uses of prostaglandin F2a analogs: beyond the eyelashes. J Am Acad Dermatol. 2015;72(4):712–716. |
||
|
Colombe L, Michelet JF, Bernard BA. Prostanoid receptors in anagen human hair follicles. Exp Dermatol. 2008;17(1):63–72. |
||
|
Zhang Q, Major MB, Takanashi S, et al. Small-molecule synergist of the Wnt/beta-catenin signaling pathway. Proc Natl Acad Sci U S A. 2007;104(18):7444–7448. |
||
|
Fong P, Tong HH, Ng KH, Lao CK, Chong CI, Chao CM. In silicoprediction of prostaglandin D2 synthase inhibitors from herbal constituents for the treatment of hair loss. J Ethnopharmacol. 2015;175:470–480. |
||
|
Weng Z, Zhang B, Asadi S, et al. Quercetin is more effective than cromolyn in blocking human mast cell cytokine release and inhibits contact dermatitis and photosensitivity in humans. PloS One. 2012;7(3):e33805. |
||
|
Norman P. Update on the status of DP2 receptor antagonists; from proof of concept through clinical failures to promising new drugs. Expert Opin Investig Drugs. 2014;23(1):55–66. |
||
|
Pettipher R. The roles of the prostaglandin D(2) receptors DP(1) and CRTH2 in promoting allergic responses. Br J Pharmacol. 2008;153(Suppl 1):S191–S199. |
||
|
Garza LA, Liu Y, Yang Z, et al. Prostaglandin D2 inhibits hair growth and is elevated in bald scalp of men with androgenetic alopecia. Sci Transl Med. 2012;4(126):126ra34. |
||
|
Kwack MH, Shin SH, Kim SR, et al. I-Ascorbic acid 2-phosphate promotes elongation of hair shafts via the secretion of insulin-like growth factor-1 from dermal papilla cells through phosphatidylinositol 3-kinase. Br J Dermatol. 2009;160(6):1157–1162. |
||
|
Harel S, Higgins CA, Cerise JE, et al. Pharmacologic inhibition of JAK-STAT signaling promotes hair growth. Sci Adv. 2015;1(9):e1500973. |
||
|
Quintás-cardama A, Vaddi K, Liu P, et al. INCB018424 : therapeutic implications for the treatment of myeloproliferative neoplasms Preclinical characterization of the selective JAK1 / 2 inhibitor INCB018424 : therapeutic implications for the treatment of myeloproliferative neoplasms. Blood. 2014;115(15):3109–3117. |
||
|
Geyer HL, Mesa RA. Therapy for myeloproliferative neoplasms: when, which agent, and how? Hematology Am Soc Hematol Educ Program. 2014;2014(1):277–286. |
||
|
Ghoreschi K, Jesson MI, Li X, et al. Modulation of innate and adaptive immune responses by tofacitinib (CP-690,550). J Immunol. 2012;186(7):4234–4243. |
||
|
Kontzias A, Laurence A, Gadina M, O’Shea JJ. Kinase inhibitors in the treatment of immune-mediated disease. F1000 Med Rep. 2012;4:5. |
||
|
Bao L, Zhang H, Chan LS. The involvement of the JAK-STAT signaling pathway in chronic inflammatory skin disease atopic dermatitis. JAKSTAT. 2013;2(3):1–8. |
||
|
Rushton DH. Nutritional factors and hair loss. Clin Exp Dermatol. 2002;27(5):396–404. |
||
|
Oshima H, Rochat A, Kedzia C, Kobayashi K, Barrandon Y. Morphogenesis and renewal of hair follicles from adult multipotent stem cells. Cell. 2001;104(2):233–245. |
||
|
Huelsken J, Vogel R, Erdmann B, Cotsarelis G, Birchmeier W. beta-Catenin controls hair follicle morphogenesis and stem cell differentiation in the skin. Cell. 2001;105(4):533–545. |
||
|
Yamauchi K, Kurosaka A. Inhibition of glycogen synthase kinase-3 enhances the expression of alkaline phosphatase and insulin-like growth factor-1 in human primary dermal papilla cell culture and maintains mouse hair bulbs in organ culture. Arch Dermatol Res. 2009;301(5):357–365. |
||
|
Sotoodehnejadnematalahi F, Burke B. Structure, function and regulation of versican: the most abundant type of proteoglycan in the extracellular matrix. Acta Med Iran. 2013;51(11):740–750. |
||
|
Xu XR, Zhang D, Oswald BE, et al. Platelets are versatile cells: new discoveries in hemostasis, thrombosis, immune responses, tumor metastasis and beyond. Crit Rev Clin Lab Sci. 2016;53(6):409–430. |
||
|
Lacci KM, Dardik A. Platelet-rich plasma: support for its use in wound healing. Yale J Biol Med. 2010;83(1):1–9. |
||
|
Mussano F, Genova T, Munaron L, Petrillo S, Erovigni F, Carossa S. Cytokine, chemokine, and growth factor profile of platelet-rich plasma. Platelets. 2016;27(5):467–471. |
||
|
Martinez-Zapata MJ, Martí-Carvajal AJ, Solà I, et al. Autologous platelet-rich plasma for treating chronic wounds. Cochrane Database Syst Rev. 2016;(5):CD006899. |
||
|
Modarressi A. Platlet Rich Plasma (PRP) improves fat grafting outcomes. World J Plast Surg. 2013;2(1):6–13. |
||
|
Gentile P, Garcovich S, Bielli A, Scioli MG, Orlandi A, Cervelli V. The Effect of platelet-rich plasma in hair regrowth: a randomized placebo-controlled trial. Stem Cells Transl Med. 2015;4(11):1317–1323. |
||
|
Lynch MD, Bashir S. Applications of platelet-rich plasma in dermatology: a critical appraisal of the literature. J Dermatolog Treat. 2016;27(3):285–289. |
||
|
Trink A, Sorbellini E, Bezzola P, et al. A randomized, double-blind, placebo- and active-controlled, half-head study to evaluate the effects of platelet-rich plasma on alopecia areata. Br J Dermatol. 2013;169(3):690–694. |
||
|
Oh Y, Kim BJ, Kim MN. Depressed facial scars successfully treated with autologous platelet-rich plasma and light-emitting diode phototherapy at 830 nm. Ann Dermatol. 2014;26(3):417–418. |
||
|
Singhal P, Agarwal S, Dhot PS, Sayal SK. Efficacy of platelet-rich plasma in treatment of androgenic alopecia. Asian J Transfus Sci. 2015;9(2):159–162. |
||
|
Valente Duarte de Sousa IC, Tosti A. New investigational drugs for androgenetic alopecia. Expert Opin Investig Drugs. 2013;22(5):573–589. |
||
|
Alves R, Grimalt R. Randomized placebo-controlled, double-blind, half-head study to assess the efficacy of platelet-rich plasma on the treatment of androgenetic alopecia. Dermatol Surg. 2016;42(4):491–497. |
||
|
Cho JW, Kim SA, Lee KS. Platelet-rich plasma induces increased expression of G1 cell cycle regulators, type I collagen, and matrix metalloproteinase-1 in human skin fibroblasts. Int J Mol Med. 2012;29(1):32–36. |
||
|
Mehta V. Platelet-rich plasma: a review of the science and possible clinical applications. Orthopedics. 2010;33(2):111. |
||
|
Leo MS, Kumar AS, Kirit R, Konathan R, Sivamani RK. Systematic review of the use of platelet-rich plasma in aesthetic dermatology. J Cosmet Dermatol. 2015;14(4):315–323. |
- Published in Corporate News / Blog
Global Stem Cells Group Announces New Business Development Director for Africa and Europe

Holya Aziza El Materi Ep Khemiri, M.D.
Global Stem Cells Group announces the appointment of Holya Aziza El Materi Ep Khemiri, M.D. as Vice President Business Development Africa and Europe. Khemiri will oversee development of a new Stem Cell Center in Tunisia.
MIAMI, Nov. 1, 2018—Global Stem Cells Group founder and CEO Benito Novas has named Holya Aziza El Materi Ep Khemiri, M.D. Vice President Business Development for Africa and Europe. The announcement is the latest update on the regenerative medicine organization’s expansion of its clinical presence on the two continents.
Khemiri, who specializes in pharmaceutical and parapharmacy medicines, product manufacturing and medical industry business development, will be in charge of developing and opening a new Stem Cell Center in Tunisia focused on the treatment of patients with degenerative illnesses and orthopedic impairments, and professional athletes with sports injuries.
Stem Cell Center Tunisia will provide regenerative medicine training to African physicians who wish to offer stem cell therapies in their practices.
Since opening the first African Stem Cell Center in Casablanca, Morocco in July, 2017, GSCG has accelerated momentum to bring regenerative medicine, training and education to Africa and Europe through expanded networking opportunities introducing the organization’s authoritative training programs, workshops, seminars, online marketplace for regenerative medicine products, expert instruction and advice on marketing for regenerative practices and other innovative initiatives.
Stem Cell Center Tunisia is part of Global Stem Cells Group’s strategy to facilitate international growth and bring quality-of-life-enhancing regenerative medicine to patients in every corner of the world. The Tunisian center’s medical team will work closely with regional patients, educators and regulators to foster engagement and promote stem cell medicine technology.
ction as a regenerative medicine reference for physicians in North Africa, providing physicians with instruction in stem cell therapies and protocols Those wishing to implement stem cell oversight in their practices will be able to visit the office and collaborate other physicians, participate in educational classes and workshops, and receive the training necessary to start implementing regenerative medicine in their practices and clinics.
Additionally, Stem Cell Center Tunisia will offer physicians a stem cell processing center, GSCG’s complete solution for onsite tissue processing, isolation, culturing and cryopreservation of stem cells. One of the biggest challenges of using traditional stem cell harvesting protocols in medical office settings is obtaining enough stem cells to treat certain degenerative conditions such as autism, lupus. Parkinson’s Disease, Alzheimer’s Disease and others.
The stem cell processing center provides a solution that allows physicians to expand stem cell colonies and obtain a significantly larger number of cells sufficient to treat patients. It also helps with patients needing multiple stem cell treatments by allowing the physician to perform a single extraction of stem cells from fat tissue; the cells harvested can then be saved and expanded, eliminating the need for new cell extraction with each treatment.
According to Novas, Khemiri’s skills and experience make her an ideal candidate to spearhead Stem Cell Center Tunisia’s development and opening.
“Dr. Khemiri brings an array of expertise and knowledge to our expansion efforts in Africa and Europe,” says Novas. She joins Global Stem Cells Group’s management team as a welcome addition at a time when our international expansion endeavors are growing exponentially.”
Khemiri was president of Tunisia’s Society Isotope Radioavtive (SISORA) from 2011 to 2014, where she was involved in manufacturing radioactive tracers to detect cancer. As president of the organization, she helped establish a partnership between SISORA and Belgium-based Radio Pharma Solultions (IBA).
From 2008 to 2011, she served as Research and Development Director of ADWYA, a Tunisia-based company specializing in the production of pharmaceutical products for medical and veterinary use and the country’s first private pharmaceutical laboratory and one of its largest private pharmaceutical companies.
In 2010, she served as creative director of RAD where she focused on formula and scientific studies and registration of medicines, skin care and food supplements with the Tunisian Health Ministry.
Khemari received her medical training at the Catholic University of Louvain in Brussels, Belgium from 1999 to 2006. She received a diploma in Pharmaceutical Sciences from the the Institut Roger Lambion in Brussels and a diploma in Biochemistry, Cosmetology, and Science Perfumery from the University Libre in Brussels.
For more information, visit the Global Stem Cells Group website, email info@stemcellsgroup.com or call 305-560-5337.
About Global Stem Cells Group:
Global Stem Cells Group (GSCG) is a worldwide network that combines seven major medical corporations. Each corporation is focused on furthering scientific and technological advancements in cutting-edge stem cell research, development, treatment, and training. The united efforts of GSCG’s affiliate companies provide medical practitioners with a one-stop epicenter for stem cell solutions that adhere to the highest medical standards.
Global stem cell’s mission is to be the largest recognized stem cell and regenerative medicine network in the world. About Adimarket: Adimarket, Inc., a division of the Global Stem Cells Group, is a one-stop, cost-competitive online marketplace for quality regenerative medicine equipment and supplies for physicians and healthcare professionals.
###
Holya Aziza El Materi Ep Khemiri named Vice President Business Development Africa and Europe.
- Published in Press Releases
Stem Cell Research and Stem Cell Therapy: When can stem cells be used to treat patients?
The difference between stem cell research and therapy is in the scientific evidence that supports therapeutic intervention to be beneficial for the patient.
Stem cells have the remarkable potential to develop into many different types of cells in the body during early life and growth. In addition, in many tissues, stem cells serve as a sort of internal repair system, dividing essentially without limit to replenish other cells as long as the individual is alive. When a stem cell divides, each new cell has the potential either to remain a stem cell or become another type of cell with a more specialized function, such as a muscle cell, a red blood cell, or a brain cell.
Stem cell research on adult stem cells
Stem cells are distinguished from other cell types by two important characteristics. First, they are unspecialized cells capable of renewing themselves through cell division, sometimes after long periods of inactivity. Second, under certain physiologic or experimental conditions, they can be induced to become tissue- or organ-specific cells with special functions. In some organs, such as the gut and bone marrow, stem cells regularly divide to repair and replace worn out or damaged tissues. In other organs, such as the pancreas and the heart, stem cells only divide under special conditions.
Until recently, scientists primarily worked with two kinds of stem cells from animals and humans: embryonic stem cells and non-embryonic “somatic” or “adult” stem cells in stem cell research.
In 2006, researchers made a breakthrough by identifying conditions that would allow some specialized adult cells to be “reprogrammed” genetically to assume a stem cell-like state. This new type of stem cell is called induced pluripotent stem cells (iPSCs).
Stem cells are important for living organisms for many reasons. In the 3- to 5-day-old embryo, called a blastocyst, the inner cells give rise to the entire body of the organism, including all of the many specialized cell types and organs such as the heart, lungs, skin, sperm, eggs and other tissues. In some adult tissues, such as bone marrow, muscle, and brain, discrete populations of adult stem cells generate replacements for cells that are lost through normal wear and tear, injury, or disease.
Stem cell research for treating disease
Given their unique regenerative abilities, stem cells offer new potentials for treating diseases such as diabetes, and heart disease. However, much work remains to be done in the laboratory and the clinic to understand how to use these cells for cell-based therapies to treat disease, which is also referred to as regenerative or reparative medicine.
Laboratory studies of stem cells enable scientists to learn about the cells’ essential properties and what makes them different from specialized cell types. Scientists are already using stem cells in the laboratory to screen new drugs and to develop model systems to study normal growth and identify the causes of birth defects.
Research on stem cells continues to advance knowledge about how an organism develops from a single cell and how healthy cells replace damaged cells in adult organisms. Stem cell research is one of the most fascinating areas of contemporary biology, but, as with many expanding fields of scientific inquiry, research on stem cells raises scientific questions as rapidly as it generates new discoveries.
In 1964, the World Medical Association developed the Declaration of Helsinki as a statement of
ethical principles for medical research involving human subjects. It includes research on identifiable human material and data, last amended in October 2013.
According to the Helsinki Declaration, in the treatment of an individual patient where proven interventions do not exist or other known interventions have been ineffective, the physician, after seeking expert advice, with informed consent from the patient or a legally authorized representative, may use an unproven intervention if in the physician’s judgement it offers hope of saving life, re-establishing health or alleviating suffering.
Intervention should subsequently be made the object of research, designed to evaluate its safety and efficacy. In all cases, new information must be recorded and, where appropriate, made publicly available.
- Published in Corporate News / Blog
Stem Cell Myths, Busted
The term stem cell research gleans different reactions from people, both in the medical community and the wider public. Still an emerging science, stem cell research is shrouded by many myths and misconceptions. Here, we take on some of the most predominant myths to discuss the misconceptions and clarify the facts regarding this fast-growing branch of medicine.
Stem cell myths
Myth #1: Stem cells only come from embryos.
FACT: False. Stem cells exist in all bodies, from embryos to adults.
Embryonic stem cells come from the early embryo, and have the potential to produce all the specialized cells of the body. Because of this, they hold great promise for studying and potentially treating disease and injuries. Tissue or “adult” stem cells are found in the body throughout our lives. These cells maintain and repair many tissues in the body. Examples of these cells include blood stem cells, muscle stem cells, bone marrow stem cells, adipose tissue (fat) stem cells and skin stem cells. Some of these adult stem cells are used in established medical and aesthetic treatments.
Myth #2: Induced pluripotent stem cells (iPSCs) eliminate the need for embryonic cells
FACT: False. Research is needed on all types of cells because it is not clear which cells will be most useful for which types of application. For the foreseeable future, side-by-side research on both embryonic and induced pluripotent stem cells is needed. Global Stem Cell Group’s research and treatment products use no embryonic stem cells.
Myth #3: Stem cell research leads to cloning humans.
FACT: False. Most countries prohibit this type of cloning.
In most countries, even attempting to clone a human being is illegal. Some countries do allow something called “therapeutic cloning” for the purposes of studying a disease. In this procedure, scientists isolate embryonic stem cells from a cloned blastocyst (early stage embryo) but do not transfer the blastocyst into a womb. In therapeutic cloning, the blastocyst is not transferred to a womb. Instead, embryonic stem cells are isolated from the cloned blastocyst. These stem cells are genetically matched to the donor organism for studying genetic disease. For example, stem cells could be generated using the nuclear transfer process described above, with the donor adult cell coming from a patient with diabetes or Alzheimer’s. The stem cells could be studied in the laboratory to help researchers understand what goes wrong in diseases like these.
Therapeutic cloning also could be used to generate cells that are genetically identical to a patient’s. A patient transplanted with these cells would not suffer the problems associated with transplant rejection. To date, no human embryonic stem cell lines have been derived using therapeutic cloning.
Myth #4: Adult stem cells are only found in adults
FACT: False. There are three different types of stem cells: embryonic stem cells, induced pluripotent stem cells and tissue specific stem cells. It’s the tissue stem cells that are often called “adult” stem cells, but these “adult” stem cells are found in people of all ages. (See myth #1).
Stem cell myths: research
Myth #5: Embryonic stem cell research is banned in Europe.
FACT: False. The laws vary across the EU.
EU member states have diverging regulatory positions on human embryonic stem cell research. For instance, in Germany, the use of embryos for research is heavily restricted under the Embryo Protection Act (Embryonenschutzgesetz) of 1991, which makes the derivation
of embryonic stem cell lines a criminal offense. But in the UK, embryonic stem cell research is allowed, subject to licensing from the Human Fertilization and Embryology Authority (HFEA). Click here for country by country overviews for more details. Under the previous two European Framework programs (FP6 and F7), as well as the current program, Horizon 2020, human embryonic stem cell research can be funded, provided that the work is permitted by law in the country where it is to take place.
Myth #6: Stem cell research and treatment is against the law in the US.
FACT: False. The FDA does not regulate the practice of medicine, but rather drugs and medical devices and which of these can be marketed in the US. Under federal law, cultured (grown) stem cell products are considered a drug, but are not illegal. Adult stem cells, however, are not cultured—they exist in our bodies throughout our organs, blood, skin, teeth, fat, bone marrow and other places.
Adult stem cell therapy is currently used in the United States to treat conditions such as leukemia and other illnesses. Bone marrow consists of stem cells which have been transplanted for years in the US.
Global Stem Cells Group offers stem cell treatments in countries where stem cell therapy is approved and regulated with no appreciable difference in safety record.. Stem cell therapy technology is still under review by the FDA.
Stem cell myths: therapies
Myth #7: Bone marrow is the best source of stem cells.
FACT: False. Bone marrow is just one source of stem cells. Bone marrow stem cells have been studied for decades, and have been used to treat certain types of cancer. A great deal of research has been dedicated to understanding this source of stem cells and their potential. Bone marrow contains a number of different kinds of stem cells, one of which is mesenchymal stem cells. However, mesanchymal stem cells can also be found in adipose (fat) tissue at nearly 2000 times the frequency of bone marrow.
Mesenchymal cells have the capability to become different types of tissues (blood vessels, muscle tissue, etc.) and are capable of communicating with other cells. In combination with other proteins, molecules and regenerative cells found in adipose tissue, they also have the ability to reduce inflammation, regenerate damaged tissue, and grow new blood vessels, a process known as angiogenesis. Stem cells from adipose tissue are more accessible and abundant. They can be processed immediately and reintroduced into the body right away.
Myth #8: There is a risk of rejection with stem cell therapy.
FACT: False. When a patient’s stem cells are derived from his or her own body (such as fat tissue), there is no risk of rejection. In fact, studies thus far have indicated no safety issues with fat-derived autologous (from self) stem cells. Since these stem cells come from your own body, the risk of rejection is eliminated.
###
- Published in Corporate News / Blog
How Stem Cell Therapies Can Help Heal Sports Injuries
Continuing our recent discussion of stem cell therapies for sports injuries, the use of mesanchysmal stem cells (MSCs) in orthopedic medicine can help in the repair of damaged tissue by harnessing the healing power of undifferentiated cells that form all other cells in our bodies. The process involves isolating these stem cells from a sample of your blood, bone marrow or adipose tissue (fat cells), and injecting it into the damaged body part to promote healing. Platelet-rich-plasma (PRP), a concentrated suspension of platelets (blood cells that cause clotting of blood) and growth factors, is also used to assist the process of repair.
Below are some examples of injuries and areas of research involving the use of mesenchymal stem cells (MSCs), which are (adult) tissue stem cells that are not only able to produce copies of themselves, but also able to divide and form bone, cartilage, muscle, and adipose (fat) stem cells when cultured under certain conditions:
Cartilage Damage
Cartilage has long bee
Recently, researchers increased their focus on the use of MSCs for treatment of cartilage damage in the knee. Some data from animal models suggest that damaged cartilage undergoes healing more efficiently when MSCs are injected into the injury, and this can be further enhanced if the MSCs are modified to produce growth factors associated with cartilage. It has been shown that once the MSCs are injected into the knee they attach themselves to the site of damage and begin to change into chondrocytes, promoting healing and repair. A small number of completed clinical trials in humans using MSCs to treat cartilage damage have reported some encouraging results, but these studies used very few patients, making it difficult to accurately interpret the results. There are currently a number of ongoing trials using larger groups of patients, and the hope is that these will provide more definite information about the role MSCs play in cartilage repair.
Tendinopathy
Tendinopathy relates to injuries that affect tendons – the long fibrous tissues that connect and transmit force from 
MSCs have the ability to generate cells called tenoblasts that mature into tenocytes. These tenocytes are responsible for producing collagen in tendons. This link between MSCs and collagen is the focus for researchers investigating how stem cells may help treat tendinopathy. Substantial research has been carried out on racehorses as they suffer from high rates of tendinopathy, and the injury is similar to that found in humans. Researchers discovered that by injecting MSCs isolated from an injured horse’s own bone marrow into the damaged tendon, recurrence rates were almost cut in half compared to horses that receive traditional medical management for this type of injury. A later study by the same group showed the MSCs improved repair, resulting in reduced stiffness of the tissue, decreased scarring and better fusion of the new fibers with the existing, undamaged tendon. It is not yet clear if these results are due to MSCs producing new tenocytes or their ability to modulate the environment around the tendinopathy, as described above. These promising results paved the way for the first pilot study in humans.
Bone Repair

Brain injury in sports
There is mounting evidence that those taking part in sports where they are exposed to repetitive trauma to the head and brain are at a higher risk of developing neurodegenerative disorders, some of which are targets for stem cell treatments. For example, it has been reported that the rate of these diseases, like Alzheimer Disease, were almost four times higher in professional American football players compared to the general population. While the cause of this disease is not yet clear, it is associated with abnormal accumulation of proteins in neural cells that eventually un
Boxers suffering from dementia pugilistica, a disease thought to result from damage to nerve cells, can also demonstrate some symptoms of Parkinson’s Disease (among others). In healthy brains, specialized nerve cells called dopaminergic neurons produce dopamine, a chemical that transmits signals to the part of the brain responsible for movement. The characteristic tremor and rigidity associated with Parkinson’s Disease is due to the loss of these dopaminergic neurons and the resulting loss of dopamine production. Researchers are able to use stem cells to generate dopaminergic neurons in the lab that are used to study the development and pathology of this disease. While a recent study reported that dopaminergic neurons derived from human embryonic stem cells improved some symptoms of the disease in mice and rats, stem cell based treatments are still in the development phase.
- Published in Corporate News / Blog


![Secondary-Diabetes[1]](https://www.stemcellsgroup.com/wp-content/uploads/2020/02/Secondary-Diabetes1-460x260_c.png)
![SCCAA_138150_F001[1]](https://www.stemcellsgroup.com/wp-content/uploads/2019/12/SCCAA_138150_F0011-460x260_c.jpg)