Which Works Better, Viscosupplementation, or Platelet Rich Plasma?
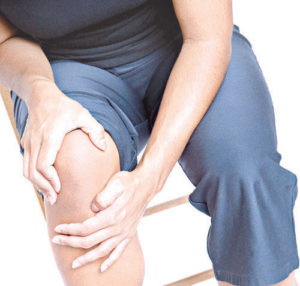
One of the treatments that the FDA approved for knee arthritis is a Hyalronic Acid (HA) injection, sometimes also known as Viscosupplementation. It has been incredibly successful for knee arthritis. In fact, so successful that many physicians are starting to use it on other parts of the body, like the hip and shoulder, which the FDA does not approve of.
HA, when injected, works like the fluid that naturally surrounds your joints. This fluid can be like a lubricant for your joints, and absorb shock, allowing bones that otherwise cause arthritis pain cause much less. Over time, it is even absorbed into the joint, which can cause the body to create a more stable cartilage all on its own.
The evidence for this treatment is astounding, with a systematic review of 76 trials, all of which were randomized controller trials. The review noted that HA, when injected, can benefit function, reduce pain, and can be a reliable and effective treatment for knee osteoarthritis.
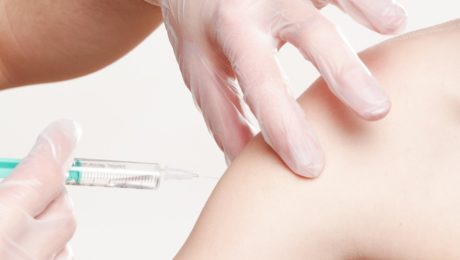
On the other hand, there is PRP therapy. Platelet Rich Plasma, or PRP, which is a type of blood that has 6 to 10 times more platelets than what is normally found in blood. They even contain many growth factors, such as Epidermal Growth Factor, Connective Tissue Growth Factor, and many more. These can help heal injured parts of the body by using the bodies natural healing tools.
However, PRP is not regulated by the FDA, and devices that are used to make PRP require said approval. Aside from this, multiple studies shoe that PRP can be very effective in the treatment of tendon injuries, as well as for osteoarthritis. This treatment can even help in the reduction of pain. There are even more studies being conducted on whether it can help other things, such as hair regrowth, cardiac muscle repair, and even dermatologic rejuvenation.
So should you use HA injections, or PRP?
In many studies, PRP has bee demonstrated to work just as well, if not better than HA. HA is also only FDA approved for the knee, meaning that it is not approved or covered for the use in any other joint. Also, the risk of infection and rejection is far less while using PRP, as it is a substance that comes from your own body, and contains white blood cells, which can help fight infection.
PRP also saves money in the long run, as using HA in a joint other than the knee is not FDA approved or covered by insurance. As a result, it can cost your patient 1500$ or more. This can even be on top of various other charges, such as doctors visits, and even the injection itself. PRP, on the other hand, only costs from 800 to 1200$ out of pocket.
So PRP has been demonstrated to be just as effective, if not better, than HA injections when it comes to arthritis pain. It does not pose a risk for infection or and auto-immune reaction either, and is even far cheaper than HA. So picking which one to use should be a no-brainer.
- Published in Corporate News / Blog
How Regenerative Microneedling Can Help Rejuvenate Skin
One of the main things that causes many people cosmetic distress and low self esteem is acne scarring. Most of us have some kind of experience when it comes down to scarring, but the most embarrassing scars are the ones that are out in the open. Such as on our faces. So what can Platelet Rich Plasma do for this?
Well, both the Department of Dermatology, Venereology, and Leprosy, as well as the National Institutes of Medical Sciences have indicated that it can be used as a viable treatment for eliminating scars. One study, done by the Department of Dermatology, Venereology, and Leprosy, sought to evaluate just how effective and safe PRP therapy can be, especially when combined with microneedling. Microneedling has been a common treatment on its own for acne scars, using distilled water, for a long time.
The way that Microneedling works is that it is used to initiate collagen synthesis on the face, thus allowing the skin to heal itself. What it does is cause micro injuries to the skin of the face using small needles. However, since the needles are so small and fine, that it does not cause any serious injury. The initiation transmits electrical signals, which calls on the body to begin a healing process, by causing small inflammation and bringing growth factors to the area.
This healing process causes new blood cells to form, and thus helps to remove the scarring over time. General treatment times are known to take from just a couple of weeks up to a year in many cases. 50 patients were involved in this study, all aged 17-32, and all suffering from acne scars.
On one side of the face, they used regular distilled water combined with the microneedling procedure, and on the right they used a topical solution of PRP. After 3 treatments giving withing the time span of a 3 months, each treatment being 1 month apart, the results that used PRP was much more likely to show improvement over the distilled water group. This would be 62.2% improvement vs 45.8% respectively. This showed that PRP can have great success in managing acne scars, and helping to make them go away.
This shows that when it comes to PRP therapy, the science again shows that there is a good and significant use for it. So what is stopping you from implementing this therapy for your patients?
- Published in Corporate News / Blog
How can PRP treat Tennis Elbow?
Despite what the name may imply, not all people who suffer from Tennis Elbow even play tennis. In fact, most of them aren’t. Many of them can be painters, butchers, plumbers, carpenters, and even much any career which can overuse the muscles in the forearm. This can cause the tendons elbow to become painful and inflamed.
The most common treatment for Tennis Elbow after the injury has taken place is rest, anti-inflammatory medicines, and generally physical therapy if it is needed. However, this is more of a temporary fix than a longstanding one. In many cases, if untreated, the pain will worsen, and many need things such as braces for their arm, injects of steroids, and shock wave therapy. Sometimes, although not often, they might even need surgery.
Most insurance companies are able to cover this surgery, as it is deemed medically nece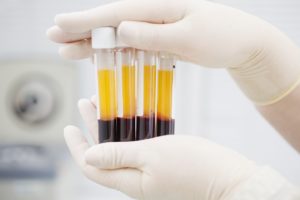
So as an alternative for surgery, perhaps try PRP, or Plasma Rich Platelet Therapy? This is a pretty simple procedure that utilizes the platelets and cells in your own body to heal your arm from the inside out. We just take out a bit of your blood, put it in a centrifuge, and extract the PRP to use on your injury. After awhile, it become good as new, without any need for surgery. It also does not need any anti-inflammatory medications, which can lower the side effects that you will see from medications and multiple surgeries.
So try it out if you would like a more holistic way of healing yourself.
- Published in Corporate News / Blog
What’s all the fuss about Regenerative Medicine?
In popular media, the term Regenerative Medicine, or Stem Cell Therapy, are becoming buzz words. This is because the field of medicine and healthcare is expanding and advancing every day, and many new treatments for otherwise common ailments are being discovered. These conditions range from burns, joint pain, strain, and pretty much every other common ailment out there
Many patients have given up hope with trying to find traditional medicines that work. This is why many people are flocking to try Regenerative Medicine. This is also something that many people who are into holistic healing are trying, as it is simply the body working to heal itself.
Regenerative Medicine works as it takes a sample of your own blood, bone marrow, and other tissues, and then it goes through a process in which to take out a certain material known as Platelet-Rich Plasma. This PRP is then applied to the infected area, so that your body’s own platelets can work to heal your body back to full health, without having to worry about any invasive surgeries.
A good question to ask is why our body does this do this itself. Well, this is because research has shown that by isolating them, they activate, and as a result when injected back into the body start to work harder to fix the issues, such as in a joint, or helping to relieve pain. Many patients who try it say they have gotten good results from the treatment.
Many doctors predict that this therapy will help physicians provide a more non-intrusive treatment that has fewer side effects, and can be big within the coming years. Many compare it to the invention of penicillin with how important it is. It is even growing in popularity with many physicians using training courses to help their patients, leaving many of them happier and healthier.
- Published in Corporate News / Blog
Platelet-Rich Plasma Vs Stem Cell Therapy: Who’s Bad?
The entire field of orthopedics is looking for new regenerative technology that can save more patients more safely. Currently there are two contenders: Platelet-Rich Plasma and Stem Cell.
While PRP is the safest of the two, it’s really hard to dismiss the remarkable capabilities of stem cell therapy. In fact, I believe it’s the future of regenerative medicine. But not at the level it’s playing right now. Which is a totally different discussion we’ll save for another day.
The thing is… there are potential harm with stem cells. And unlike PRP, stem cell’s constituents are man-made, so things can go wrong. We’ll discuss the potential dark side of this therapy later in this article. However, I feel it’s important to highlight how good a treatment stem cell therapy is.
Quick Overview: Stem Cell Vs Platelet-Rich Plasma
Platelet-Rich Plasma is like water and nutrients that help restore (and sometimes accelerate) your body’s EXISTING healing mechanism. If your body is stuck with its healing, PRP can help. It releases growth factors and cytokines to kick start the healing. Stem cells on the other hand is not used to enhance healing, but to create new solutions to healing challenges. So it’s more for tissues that are totally lost.
Stem Cell Vs Platelet-Rich Plasma
With me? Before we proceed, let’s look at a little background of stem cells. We’ll stick to orthopedics for the sake of simplicity.
Orthopedic Stem Cell Therapy
Stem cells are naturally found in the human body and they are a fundamental part of the body’s normal healing process. Stem cells are known as ‘raw potential’ as they can be converted into any cell that the body needs. The body utilizes stem cells to substitute damaged and/or injured cells. This process allows natural healing and repair of the injured or damaged cells.
As the body gets older the amount of natural reserved stem cells starts to decline, which explains why the healing process is slower as the body gets older. Stem cell therapy resolves this shortage by injecting supplementary stem cells into the injured/damaged area of the body, which triggers the cell replacement, natural healing, and pain relief.
Stem cell therapy is a simple and quick procedure, taking about 15 minutes. Pain discomfort is often felt immediately, with the majority people reporting a significant improvement within one to two days.
With stem cell therapy the patient does not have to have any type of surgical procedure, local or general or downtime. Most of the patients experience a complete restoration of the damaged/ injured ligaments, tendons, and cartilage within about in 28 days. Stem cell therapy has been proven to be complexly safe, with no side effects reported in the US or in Europe.
The Difference Between Stem Cell Therapy and Platelet-Rich Plasma (PRP) Therapy
Often times, stem cell therapy and PRP can be confused because they have a lot in common during the healing process. The easiest way to tell the difference between the two, is PRP is removed from the patient’s own body, it goes through a scientific process and is them injected into the area being treated.
The cells used for stem cell therapy can come from a few different places; from an unviable embryo, and unviable fetal stem cells these stem cells are the most often used because the cells are unspecialized and can be made into specialized cells. As it sounds, preparing stem cells for therapy is a complex process. Stem cells are produced in a sophisticated labs by cell biologists and are typically grown over several weeks before it’s ready.
Plus, adult stem cells may be used, although it is not nearly as common yet because scientists are still working on ways to identify stem cells within the tissue of an adult human body.
Stem Cell Vs Platelet-Rich Plasma
So what’s the dark side of Stem Cell Therapy?
The obvious concern is that treatments with stem cells could be dangerous if not carefully controlled. I know we are all doing things for saving lives and helping people live longer, more happily, but the risks must also be considered.
Below are the 5 risks that stem cells carry. (which Platelet-Rich Plasma doesn’t.)
Risk of viruses: Since the stem cells are foreign bodies, if they happen to carry harmful microscopic agents, it’ll bring unnecessary complications. Especially those patients whose immune systems are weak, could be highly vulnerable diseases.
Uncontrolled growth: As I said before, stem cells are produced in a lab and grown over a period of several weeks. However, there is very tiny possibility the growth will continue uncontrolled after installing it into the patient. We pray it doesn’t happen.
Multi-tasking of cells: Stem cells are cultivated and grown into specialized cells that are designed to be doing just one thing and one thing only. But what if, in the long run, they also do other things that wasn’t in the original scope of things? Something to ponder.
That said, I still believe stem cells hold great promise. Now, I want to take this rest of the article to highlight a few of the common conditions that are found to be best for stem cell treatments.
Stem Cell Vs Platelet-Rich Plasma
Rheumatoid Arthritis
Rheumatoid Arthritis is caused by inflammation of the joints as a result of an autoimmune progression. The body’s immune system attacks the joints. Patients with Rheumatoid Arthritis suffer from mild to severe pain, constant fatigue, warm, and swollen joints. This type of chronic inflammation has the potential to easily damage the joints. Therefore, treatment is concentrated on decreasing the inflammation and slowing down the progress of the condition. Stem cell therapy provides a treatment alternative that takes advantage of the healing and anti-inflammatory effects.
Osteoarthritis
Osteoarthritis is joint inflammation caused by the deterioration of the cartilage that cause the bones to rub up against one another. Patients who suffer from osteoarthritis have pain, stiffness, and a decrease in their range of motion in their joints. Although, there is no cure for osteoarthritis, stem cell treatment focuses on reducing the pain reduction through medication, physical therapy, or occupational therapy. Stem cell therapy provides a treatment alternative that takes advantage of the healing and anti-inflammatory effects. While medication helps with the pain.
Shoulder Repair
Shoulder injuries such as rotator cuff tears and arthritis of the shoulder joint, as well as other types shoulder pain may be responsive to stem cell therapy. Stem cells goal is to renew damaged joints.
Stem Cell Treatment for Joint Repair
Hand and elbow problems caused by arthritis of the joints is a type of deteriorating joint disease that has disabled millions of people. Definite types of wrist and elbow joint issues including certain ligamentous injuries and tendon problems may not benefit from cell therapy. It is very important that the doctor evaluate each patient to see if stem cell therapy is a viable treatment for their patients.
Stem Cell Treatment for Knee
Knee arthritis is a type of deteriorating joint disease, which affects millions of people. Most people believe there only option for pain relief and better mobility is steroid injections or surgery, including total knee replacement surgery. However, that is not the case, many people benefit greatly from stem cell therapy. Specific types of knee issues such as, ligamentous injuries and substantial meniscal injuries may not be responsive to regenerative therapy (stem cell therapy). Each case must be carefully evaluated and the orthopedist will decide what options are best for the patient, in some cases, stem cell therapy is tried even if the patient is not exactly an ideal candidate, but trying is better than just scheduling surgery.
Stem Cell Treatment for Hip
Hip arthritis is similar to knee arthritis; millions of people suffer from hip problems. Patients usually try to delay the hip replacement surgery as long as they can and try other methods such as steroid injections, which for some people do help for a short period of time. However, long tern injects can damage the tissue near the hip. While fractured hips and certain kinds of hip injuries cannot be treated with stem cell therapy, surgery is the only available option left.
Stem Cell Treatment for Joint Repair
Problems with the hands and elbow joints usually respond well to stem cell therapy. If there are problems with the ligaments and tendons, then surgery may be necessary.
Degenerative joint diseases disable millions of people. While certain types of injuries are not a good match for stem cell therapy, there are several that are a good match. Before you prescribe surgery to repair damaged or injured joints consider about stem cell therapy, and if possible give it a try first.
- Published in Corporate News / Blog
Platelet-Rich Plasma Stays Quietly Popular Despite Neglect
Fact: According to research, PRP treatments are one of the most in-demand treatments available in healthcare.
This is impressive considering the following.
PRP is not supported by the medical industry. No big pharma funding on extensive research or marketing. No medical associations lobbying to increase its awareness.
PRP is shunned by the insurance companies. No reimbursements from them. So getting patients to pay is difficult. Especially for a treatment that’s relatively “unproven” like this.
The cost of PRP treatments are actually rising. In 2006, you can get a PRP treatment for $450. Today it costs $800. The cheapest we’ve seen is $650. The prices are still robust as demand keeps up.
However, we believe the best of PRP is not even here yet. We’re just one breakthrough study away from exploding into mainstream hospitals and clinics. We see the biggest growth in Platelet-Rich Plasma happening in Asia.
Strongly based on fundamental healing theory
The growth can be attributed to PRP’s fundamental healing property. More platelets. More growth factors and cytokines. And therefore more healing. It’s as simple as that. And no one can argue this fact.
Our body’s natural healing mechanism operates with 150,000/ul-350,000/ul platelets in blood. Using Platelet-Rich Plasma means this number is amplified by 3X to 5X. How can this be not translated into better healing?
Believe it or not, the best orthopedic doctors use Platelet-Rich Plasma. And do so regularly.
PLATELET-RICH PLASMA TRENDS
PRP can be used to promote healing of injured tendons, ligaments, muscles, and joints, can be applied to various musculoskeletal problems. And they conduct regular studies to test it’s effectiveness.
One landmark study involved double-blind randomized controlled trials to see the effect of PRP on patients with chronic low back pain caused by torn discs. The study outcome says 60% of the patients felt significant improvements.
Some were cured. CURED!
Platelet-Rich Plasma Variants
So far, there are the following type of PRP variants.
Plasma Rich in Growth Factors (PRGF)
Plasma Rich in Platelets and Growth Factors (PRPGF)
Platelet-Rich Plasma (PRP); Platelet Poor Plasma (PPP)
Plasma Rich in Platelets and Rich in Leukocytes (LR-PRP)
Plasma Rich in Platelets and Poor in Leukocytes (LP-PRP)
Platelet-Rich Fibrin Matrix (PRFM)
All of them involve Plasmapherisis — the two stage centrifugation process to separate platelets from blood. However, what happen what happens after that can be different. And the industry hasn’t found it’s middle ground as to which variant to be standardized. We believe the confusion will clear up in 3-5 years.
PLATELET-RICH PLASMA TRENDS
No matter which variant you end up using, the bio-factors at play are the following:
Growth factors: TGF-B, PDGF, IGF-I,II, FGF, EGF, VEGF, ECGF
Adhesive proteins: Fibrinogen, Fibronectin, Vitronectin, Thrombospondin-1
Clotting & Anti-Clotting factors: Proteins, Antithrombin, Plasminogen, Proteases, Antiproteases
How Platelet-Rich Plasma Actually Work
Why is the treatment commonly used for wound healing and pain management? The answer is because the platelets’ main job is to aid coagulation, act as a biological glue and support stem or primary cell migration. In addition, it also helps in restoring hyaluronic acid and accelerates the synthesis of collagen and glycosaminoglycans and increases cartilage matrix.
Not only that, the platelets are delivered in a clot which means it can immediately act as a scaffold to enable the healing process. 95% of the bio-active proteins are released within 1 hour of injecting Platelet-Rich Plasma. The platelets continue to release growth factors for 7-10 days. Thus it’s recommended to re-inject PRP every 7 days.
PLATELET-RICH PLASMA TRENDS
Why are patients coughing up their hard earned money for this?
This reminds me of hundreds of thousands of PRP treatments paid from patient’s own pocket even though they’ve been paying for years to get covered by their respective insurance provider. In 2015, PRP costs were anywhere between $600 and $800 per site per treatment. And most patients go for repeated treatments. So why were they forking up their hard earned money if the treatment was not working? Weren’t there any better alternatives under the “coverage” of their insurance provider? The answer is 1) the treatment works. 2) there’s nothing else out there that’s as natural and side-effect-free as PRP.
Consider the case of osteoarthritis. 27 millions Americans are impacted by it. 33.6% of people older than 65 are victims. All of them experience gradual degeneration of cartilage and bones — they lose roughly 5% cartilage per year. Yet, our medical industry doesn’t have a fix to stop it.
However, when doctors started doing PRP treatments for their osteoarthritis patients, they found a large majority of them had no further cartilage loss.
To me, it means we should make PRP treatments the default first-line treatment for osteoarthritis across the country.
Another huge market is hair loss and cosmetic facial applications. I know there are many people who believe PRP doesn’t work for hair. Here’s what one of the Platelet-Rich Plasma studies found were the effect of the treatment on hair loss.
“Hair loss reduced and at 3 months it reached normal levels. Hair density reached a peak at 3 months (170.70 ± 37.81, P < 0.001). At 6 months and at 1 year, it was significantly increased, 156.25 ± 37.75 (P < 0.001) and 153.70 ± 39.92 (P < 0.001) respectively, comparing to baseline. Patients were satisfied with a mean result rating of 7.1 on a scale of 1-10. No remarkable adverse effects were noted.”
I’ll take that.
That’s me getting PRP for hair. ??
PLATELET-RICH PLASMA TRENDS
PRP market is expected to hit $126 million in 2016
That number looks paltry. But that’s an 180% increase over the 2009 figure of $45 million.
Consider this. Just for osteoarthritis alone, if all the 27 million Americans receive 1 PRP shot a year at a conservative $400 per treatment, it would be a market of $10 billion. And that’s one condition out of the many that Platelet-Rich Plasma injections are proven to work.
Another condition that PRP is known to work very well is Tennis Elbow. It affects on average 1% to 3% of the overall population. That number is as high as 50% among tennis players.
Do the math.
Just getting Platelet-Rich Plasma covered by insurance will unleash the market big time and will help heal millions of patients naturally, more effectively.
Oh ya, that means the insurance companies will have to pay more. Why would they?
HOWEVER, if this treatment could reduce further expensive intervention like surgery then it may actually be a blessing for the insurance guys in terms of savings. One surgery avoided by a patient through right intervention through PRP treatments will save the insurance companies at least $25,000. Now, that’s a win-win for both patients and insurance.
I believe it’s a matter of time before insurance companies start realizing their folly of not supporting this treatment.
PLATELET-RICH PLASMA TRENDS
After all is said and done, it’s still “unproven”
The problem with PRP is that it can be used for just about everything, which is a good problem to have until health care officials (and insurance companies) start realizing that people are going to misuse it.
So it’s classified as unproven. The VAST scope of the treatment calls for urgent structure and guidelines. There are some 20+ conditions where researchers have found it “helps” in one way or another. It’s a daunting task to prove its efficiency in all the areas. Nevertheless, we’ll get there.
Though we’ll need a lot of funding for that.
And yes, we need to standardize the procedure. As well as come up with optimized protocols for each conditions. Someone need to take initiative on that. We’re counting on independent doctors and medical institutions. The big pharma won’t jump in because what’s in it for them, right?
It’s so simple, you’d be an idiot to not try it.
You only need a vacuum blood harvesting tube like what we offer here, a centrifuge with adapter for the tube, pipettes and 10ml ampules of 10% calcium chloride.
The only complexity comes from not following a standard PRP system. Because the final platelet count can depend on a variety of factors. Like initial volume of blood, the technique used and relative concentration of WBC and/or RBC. As well as on the patient’s side, there are factors such as age, growth factor and WBC content.
However, concentration-wise, there’s little confusion as once a sufficiently high range is reached, more doesn’t have any adverse or enhancing effect — it saturates at a certain point. So that’s the minimum. Once you reach that, you’re good. Although the outcome is not always guaranteed to be same, with the right number of platelets, platelet activation and cytokine release, you can get a consistency in your PRP offerings.
There’s still some uncertainty over the number of injections, the timing and delivery method of Platelet-Rich Plasma. But with wide-spread adoption, some kind of structure will emerge.
Let’s hope the first glimpses of it will arrive this year.
Do you know in 2015, the world saw approximately 1 million knee arthroplasties for osteoarthritis? At $25,000 apiece, $25 billion.
How many of these patients had the good fortune of their doctor recommending PRP early on?
- Published in Corporate News / Blog
The Growth Factor Showdown: Plasma Vs Fibrin
Yep, it’s Platelet-Rich Plasma. There has been numerous speculations about which one among the latest Platelet-Rich family was the greatest—is it the plasma or the fibrin or even latest the A-fibrin? That confusion is somewhat over now.
Platelet-products are known to facilitate angiogenesis, hemostasis, osteogenesis, and bone growth. But see, the only reason plasma can do that is because of the growth factors it carries. Let’s review the specific roles of these growth factors in the healing process.
Growth Factors In Platelet-Rich Plasma
These are growth factors that are traditionally known to have played a vital healing role in PRP. If you’re seeing your patients get better as a result of that injection you gave, these are guys you need to thank for.
Platelet-Derived Growth Factor (PDGF): Regulates cell growth and division. Especially in blood vessels. In other words, this guy is the reason the blood vessels in our body reproduces.
Transforming Growth Factor Beta(TGF-b): Responsible for overall cell proliferation, differentiation, and other functions.
Fibroblast Growth Factor (FGF): Plays a vital role in the wound healing process and embryonic development. Also behind the proliferation and differentiation of certain specialized cells and tissues.
Vascular Endothelial Growth Factor: Responsible for vasculogenesis and angiogenesis. Restores oxygen supply in cells when inadequate. It also helps create new blood vessels after injury.
Keratinocyte Growth Factor (KGF): Found in the epithelialization-phase of wound healing. In other words, it causes the formation of epithelium immediately after a wound or injury occurs.
Connective Tissue Growth Factor: Major functions in cell adhesion, migration, proliferation, angiogenesis, skeletal development, and tissue wound repair.
These growth factors are what enables a Platelet-Rich product in tissue regeneration.
Platelet-Rich Plasma Rules
However, this new study suggests Platelet-Rich Plasma and it’s gelled cousin Platelet-Rich Fibrin both differ in the release of these growth factors which can significantly affect the healing outcome.
Here’s the takeaway:
“The advantage of PRP is the release of significantly higher proteins at earlier time points whereas PRF displayed a continual and steady release of growth factors over a 10-day period.”
Some argue that PRP enriched with large number of growth factors (a portion of it may even be excess) produce short-term effect and so is less desirable than a PRF whose release is slower and thus more beneficial in the long run.
That being said, PRF do have some advantage over PRP. Mainly:
It doesn’t need thrombin and anticoagulants.
It results in better healing due to its slow polymerization process.
And it helps in hemostasis.
How Platelet-Rich Plasma Differs From Platelet-Rich Fibrin
Platelet-Rich Plasma is a result of double spin method — a hard spin to separate red blood cells from everything everything else in the autologous (or whole) blood and a soft spin to separate the platelets and white blood cells. The result is Platelet-Rich Plasma (PRP), Platelet-Poor Plasma (PPP) and Red Blood Cells.
PRF is a newer method. Here after the first centrifugation, the middle layer is taken—which contains less platelets but more clotting factors. This gradually forms into a fibrin network and traps in the cytokines. It is then centrifuged in a PRF centrifuge resulting in PRF, a fibrin layer containing platelets and plasma.
What Matters In Healing
Obviously, when it comes to accelerating healing, immediate availability of growth factors and cytokines matter. So I believe PRP does a better job in this than PRF. Also the immediate release of growth factors for PRP means we can repeat the PRP injections for more healing factors just days after initial injection.
Platelet-derived products are in it’s infancy now. However, considering the huge potential benefits, there’s still a lot more research to be done. How about you? Which of these do you find beneficial?
If you’re a physician using any or both of these, do write to us and let us know of your experiences. Use the contact form here.
- Published in Corporate News / Blog
Platelet-Rich Plasma Injections: Protocol Guide
Almost all sports medicine doctors would agree that there’s no harm in trying Platelet Rich Plasma Injections (PRP Injections) for their patients. After all, there are hundreds of thousands of cases of positive results. All it needs is research to prove it’s worth. Currently there are many independent researches going on from private funding like the one conducted by Dr. Kimberly G. Harmon M.D., director of the Primary Care Sports Medicine fellowship at University of Washington. She just recently received a gift to support her research from UW alumni who I’m guessing firmly believes in Platelet-Rich Plasma (PRP).
While the process of extracting PRP is fairly simple — there are many variants as long as platelets are above baseline levels with at least seven growth factors — many physician are still unsure about what they can and can’t do when it comes to this marvelous procedure. So today I want to take the time to shine light on the fine print.
Platelet-Rich Plasma Injections Protocol
PRP: Protocols, Technique and Safety Endorsements
Protocol/Technique
Usually, the procedure requires the physician/surgeon and an assistant or two to help with the preparation of graft, the maintenance of sterile technique and saving the ultrasound images (if relevant).
Pre-Procedure Considerations
There should always be a specific indication associated with a physical exam with confirmed imaging studies such as an ultrasound, Cat Scan, or an MRI before treatment.
Proper patient education and a discussion must be had with the patient as well as a signed informed consent prior to the procedure.
Contraindications reviewed prior to procedure.
Graft Preparation
The patient is to positioned in a comfortable seated or reclining position.
Sterile single needles and syringes must be used with proper handling and disposal.
Using an aseptic procedure, the proper amount of blood is then drawn from the vein for the PRP procedure.
If the blood cannot be obtained from the site the first, time a new site must be used to prevent early activation.
Using a sterile technique, transfer the tube of venous blood to the centrifuge. Platelet Rich Plasma should be acquired using a separating device created for autologous blood. Preference is always given to a closed system that will prevent exposure of the blood and its cellular modules to the open air, and permits minimal use of the tissue.
Image Guidance PRP Therapy
Real time imaging guidance using ultrasound CT, or fluoroscopy should always be used when performing a PRP injection.
If ultrasound is going to be used, the subsequent considerations need to be decided on in advance: For lengthy procedures, PRP injections near the spine and intra-articular injections sterile gel is recommended.
Always use sterile probe covers. Cleansing the probe before and after the PRP procedures and observance to sterile technique is sufficient.
Guided images and ineradicable markings of the site of the probe position and the needle entry always needs to be made before cleaning the skin where the probe and needle will be inserted.
Always apply a bandage or a dressing after the procedure to protect the entry site from germs.
Post-Injection
The patient should be monitored for any post PRP procedure complications such as vaso-vagal.
The patients should be given their post procedure directions and precautions and any questions should be answered before they leave, they should also have emergency contact information.
Patients should also be instructed about the immobilization and any post procedure activity that is allowed and/or not allowed.
Post PRP procedure pain prescriptions need to be given to the patient before discharge and any questions they may have about the medication(s) should be answered at this time. The patient also needs to be instructed to avoid NSAIDs till they have healed, are pain free, has full function has returned to the area being treated (or at least to the limited area being treated.
Per OSHA guidelines contaminated areas must be disinfected, before the next patient uses the room (area.)
The PRP procedure must be documented in detail, which includes a procedure note that contains the following information: date, pre and post procedure diagnosis, name of the procedure, physician/surgeon(s), any assistants, whether or not anesthesia was used, and if so what type, short-term indication of the procedure, a description of the graft preparation, a description of the procedure that includes any/all guidance and instruments used.
Platelet-Rich Plasma Injections Protocol
Follow-up
Patients are normally re-examined in 2-6 weeks after the PRP procedure to follow-up on pain, use, the injection site and to discuss any concerns and any future course of action.
The patient response of the treatment should be recorded using authenticated outcome measures.
Any complications responses and all other relevant information should be logged into in the ICMS tracking system.
The consideration for another PRP injection should be the center of the discussion and the patient will be able to make a decision based on the outcome.
Safety
With every medical procedure universal precautions must be used including before, during and after the procedure.
Risk of infection – PRP is antimicrobial and provides effective protection against most bacterial infections except for Klebsiella, Pseudomonas, and Enterococcus.
With the graft being made entirely out of autologous it basically eliminates the apprehension for the transmission of disease unless the graft became contaminated.
Risks to Patient from the Procedure
Infection
Bleeding
Nerve damage
Pain
Lack of result
Loss of limb and death are very rare but possible.
Platelet Rich Plasma: Indications
Musculoskeletal complaints, require a complete history and exam to find a diagnosis. Often times, diagnostic studies may be needed and reviewed to understand why prior treatments failed. PRP is usually considered an optional treatment for chronic and subacute conditions. Commonly, healing slows down or stops all together at the 6-12 weeks’ period following an acute or traumatic injury. If the patient has not had any improvement for over the first six weeks, it’s probable the healing period has stopped.
Platelet-Rich Plasma Injections Protocol
Platelet Rich Plasma: Contraindications
Septicemia
Platelet dysfunction syndrome
Localized infection at the procedure site
Hemodynamic instability
Critical thrombocytopenia
Patient not willing to take the risks involved with the procedure
Relative Contraindications:
Regular use of NSAIDs within 48 hours of the PRP procedure
HGB of < 10 g/dl
Platelet count of < 105/ul
Systemic use of corticosteroids within 2 weeks
Recent illness or fever
Cancer – particularly hematopoietic or of the bone
HGB < 10 g/dl • Platelet count < 105/ul
Corticosteroid injection at treatment site within 1 month
Tobacco use
- Published in Corporate News / Blog
Platelet-Rich Plasma For Breast Augmentation: How it works
“You start out happy that you have no hips or boobs. All of a sudden you get them, and it feels sloppy. Then just when you start liking them, they start drooping.”
Cindy Crawford
Just like men associate (some of) their masculinity with the shape and size of their muscles, women associate (some of) their femininity with the shape and size of their breasts. However, unlike the muscles, exercise won’t be of much help for augmenting the size of breasts.
Fortunately, we have an array of procedures to the rescue. And today, we’re going to take a look at everything that Platelet-Rich Plasma can do for breast augmentation.
Platelet-Rich Plasma For Breast Augmentation
PRP & Breasts: The Incorrect Perception
Currently the traditional breast augmentation procedures like breast implant surgery and fat grafting are still the most effective methods. However, the general public do talk about Platelet-Rich Plasma for breast augmentation. And often times, they have a wrong perception of it. Here’s their typical conversation with a dermatologist goes.
Patient:”Hey, I heard about this thing called PRP, and I was told it’s just a couple of injections with stuff drawn from our own blood.”
Doctor: “Yeah, they are really good.”
Patient: “Really? You think so? I also heard they’re good for breast augmentation. Can you do it for me?”
This follows by the doctor slapping on their forehead. Then the doctor patiently explains how PRP is a healing tool and not an implant tool.
Platelet-Rich Plasma For Breast Augmentation
How Platelet-Rich Plasma For Breast Augmentation Works
Here are two ways PRP is used for breast procedures.
- Fat Transfer & Platelet Rich Plasma For Breast Lift
Fat transfer is the process of taking unwanted body fat (liposuction procedure) from other parts of the body and processing it before injecting it to upper part of the breast and in the cleavage area. This is immediate enhancement. And since it’s immediate, the sudden expansion of the breast can cause blood vessels to be blocked causing some parts of the breasts or the nipples to lose sensitivity. Sometimes it can even cause the skin at those areas to go haywire.
So the best way to avoid that is to make sure enough collagen and growth factors are supplied, well in excess of the area’s needs. That’s why it makes sense to combine the Fat Transfer procedure with Platelet-Rich Plasma. In this combination, the doctor adds PRP (Platelet Rich Plasma) derived from the patient’s own blood, to the fat when processing the fat, which includes many blood-derived growth factors and tissues containing collagen for skin rejuvenation. The end-product is then injected like a typical PRP Injection. The result is firmer breasts with not just a change in size, but also changes in skin texture and shape of the breast. And there’s no worry of losing sensitivity. Some call the entire procedure as Platelet-Rich Plasma Facelift. Results generally last from 9-18 months.
This great procedure has boosted the confidence levels of thousands of women who wanted to overcome their unnatural shaping and aging of breasts. However, even though it works for all kind of breast sizes, it’s not recommended for women with:
Extreme Loss of Volume
Excessive Sagging
Previous Breast Implants
- Only Platelet Rich Plasma For Breast Rejuvenation
This second procedure is purely PRP for rejuvenation purposes. It’s for women who’re happy with their breast size but would love to rejuvenate the skin for youthful looks, restore fullness for healthy breasts and regain sensitivity in areas where it’s diminished. The procedure is same as any other Platelet-Rich Plasma procedures. It starts with drawing 20ml of patient’s blood, spinning it (twice) in a tabletop centrifuge and then injecting to necessary areas. PRP injection not only enhances the looks, it actually produces new tissues in the area because of all that growth factors resulting in better cleavage and fullness.
So if any of your beautiful, intelligent and man-loving female patients need a little help in augmentation, you can confidently recommend these two Platelet-Rich Plasma procedures for breasts. It works.
Additional perks
Some of us in the medical profession hold the opinion that, “if you want to enlarge your breasts, stick with the gold standard. Breast Implants performed by a board certified plastic surgeon.” Artificial implants are anything but gold standard. Natural is the new gold. Besides, to implant artificial stuff you need to cut up the breasts. And the scars that results can take time to heal. Plus, implants may need to be replaced sometime after 10 or 20 years.
“It wasn’t just her beauty. It was the attitude in her smile, the tilt of her head, and the loving look in her eyes when she caught me sneaking a peek down her shirt.”
John L. Monk, Kick
- Published in Corporate News / Blog
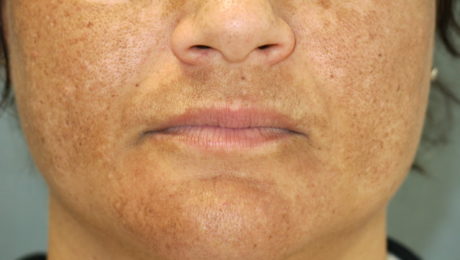
Platelet-Rich Plasma For Melasma — Will It Fade Forever?
For most women, a tiny pimple on the face is enough to ruin their day. Or week. Even the slightest imperfection that may have a 1% chance of getting noticed by others will freak them out. For these women, Melasma is their darkest nightmare. It’s a pretty common issue, a result of exposure to sun, that causes brown patches on the face. Permanent patches, I should add.
If you’re suffering from Melasma, the road to “recovery” usually looks like this.
- You hope that it’ll fade away.
- Your friend suggests you try apple cider vinegar and lemon juice treatment.
- Slightly disappointed.
- You visit a dermatologist who’ll prescribe a bleaching cream (hydroquinone or similar).
- Full-on disappointment.
- You Google the hell out of the topic.
- Overwhelm.
- Concealers and makeup becomes your best friend.
At this point, no one can convince you there is a treatment for getting rid of melasma. Trying more and more treatment only runs the risk of making the condition worse. So what would you do?
Platelet-Rich Plasma For Melasma
What about Platelet-Rich Plasma For Melasma?
According to recent Turkish and Malaysian studies, Platelet-Rich Plasma is showing great promise for melasma. The one good thing about PRP for Melasma is the fact that PRP won’t make the condition worse unlike IPL, fraxel or other treatments. So that’s one of the treatment you can confidently try without worry. It’s like getting a natural facial treatment that has a whole lot of potential benefits even if it didn’t help cure melasma.
PRP injections work by supplying growth factors to reduce the pigmentation. And being an independant treatment with no downtime, it can be done in conjunction with conventional treatments for melasma to add and enhance the effects. There are more than 30 bioactive substances in Platelet-Rich Plasma that has separate roles like increasing skin volume and adding new blood vessels to name a few.
Platelet-Rich Plasma with Microneedling
This is the most common combination for Platelet-Rich Plasma therapy. Here’s a video of Dr. Michael Somenek performing PRP injection on a patient of his immediately after microneedling. The combination is known to have produced results for a lot of varieties of skin pigmentation issues that it’d not be wise for anyone to ignore it for melasma, especially when creams and peels didn’t help. More important is PRP’s ability to stimulate collagen production in the area so it tightens the pores and makes your skin glowing.
Why Platelet-Rich Plasma?
PRP is primarily a healing vehicle. It needs to be injected into the membrane below the skin. The way it works is by supplying the underlying skin membrane with collagen and tenascin stimulated by the transforming growth factors in PRP. These growth factors also promote formation of new blood vessels that in some cases results in disappearance of spider veins.
The released growth factors (mainly platelet derived growth factor (PDGF), epidermal growth factor (EGF), vascular endothelial growth factor (VEGF), fibroblast growth factor (FGF) and transforming growth factor-beta (TGF-ß)) can stimulate proliferation of fibroblast and epidermal cell, and collagen synthesis. In addition, the transforming growth factor-beta (TGF-ß) has been proven to inhibit melanogenesis — or reverse skin pigmentation — the exact opposite effect of exposure to UV-B radiation.
Typically, patients see excellent results with 2-3 PRP injections in the first 3 months. And clinical studies have shown that it will maintain after 6 months.
However, Melasma is known to recur even after successful treatments. So you must take precautions against it by using sunscreen with broad-spectrum protection and an SPF of 30 or higher. And avoid skin care products that are harsh as they can exacerbate melasma.
- Published in Corporate News / Blog