Q&A: What are exosomes, exactly?
by James R. Edgar
Abstract
What is the current definition of an exosome?
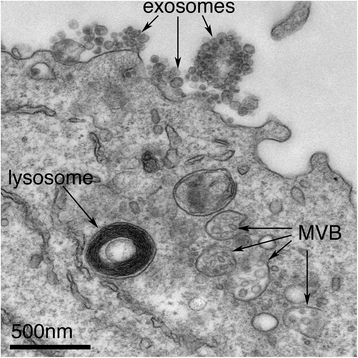
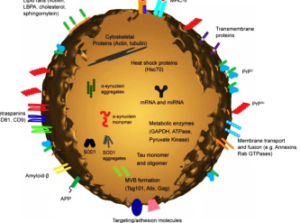
Exosomes correspond to intraluminal vesicles of multivesicular bodies. A transmission electron micrograph of an Epstein–Barr virus-transformed B cell displaying newly expelled exosomes at the plasma membrane. Multivesicular bodies (MVB) can be seen which can deliver content to lysosomes for degradation or can fuse with the cell surface to release intraluminal vesicles as exosomes, indicated by the arrows at the top of the picture
There are other types of microvesicle, including apoptotic bodies and ectosomes, which are derived from cells undergoing apoptosis and plasma membrane shedding, respectively. Although apoptotic bodies, ectosomes and exosomes are all roughly the same size (typically 40–100 nm) and all also contain ‘gulps’ of cytosol, they are different species of vesicles and understanding differences between them is of paramount importance but has too often been overlooked.
How were exosomes first recognized as distinct entities?
Even then, however, these extracellular vesicles were largely ignored, forgotten or, again, dismissed as a means of cellular waste disposal. It is only in the past decade that interest in exosomes has exploded, with a nearly tenfold increase in publications in as many years (115 in 2006, 1010 in 2015).
Why this explosion of interest?
Yet despite 20 years of research, the very basics of exosome biology are in their infancy and we know little of the part they play in normal cellular physiology.
So do we know how they are generated?
ILVs (and thus exosomes) can be generated at the endosomal limiting membrane by at least two mechanisms, one of which depends on the ESCRT machinery (ESCRT stands for endosomal sorting complexes required for transport) whereas the other is ESCRT-independent (Fig. 2).
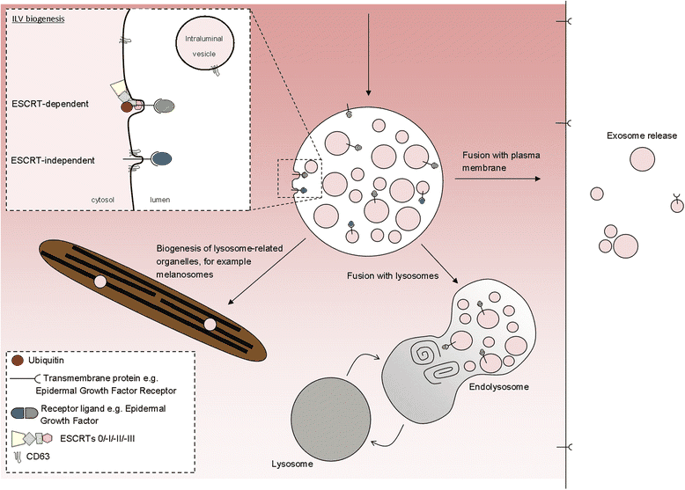
ILVs are generated by invagination of the endosomal membrane and have three possible fates. Inset: intraluminal vesicles (ILV) are formed by invagination of the endosomal membrane by either ESCRT-dependent or ESCRT-independent mechanisms. Matured endosomes accumulate ILVs within their lumen and have three distinct fates. They may deliver content that contributes to the biogenesis of specialized lysosome-related organelles (for example, melanosomes, Weibel-Palade bodies, azurophilic granules), they may fuse with lysosomes or they may fuse with the plasma membrane where released ILVs are now termed ‘exosomes’
But ILVs are still able to form in the absence of ESCRTs [8], so other means of generating ILVs must exist, although the mechanisms for their generation are less clear. Generation of these ESCRT-independent ILVs requires the tetraspanin CD63—a protein abundant on ILVs but with unclear function [9]—and may be facilitated by cone-shaped bending properties of lipids such as ceramide [10].
If not all ILVs become exosomes, what determines the fate of an ILV?
But what regulates the balance between exosome release and alternative fates of ILVs remains engimatic.
What about differences between cells: do all cells release exosomes?
Some cells—for example, the B cells, dendritic cells and mast cells of the immune system—appear to release exosomes constitutively; in fact, most of the data we have on exosomes comes from immune cells. As well as releasing exosomes constitutively, these cells may also be stimulated to secrete exosomes by cellular interactions. For example, murine dendritic cells, which are specialized to activate T lymphocytes, secrete higher levels of exosomes upon interaction with antigen-specific CD4+ T lymphocytes [13]. In fact, lymphocyte interactions generally can be accompanied by exosome release; human T cells (including primary T cells from blood, T cell clones and Jurkat cell lines) release exosomes upon activation of their antigen receptors [14] and B cells release more exosomes upon engagement with antigen-specific CD4+ T cells [15].
Other cell types can be pushed to secrete exosomes by means of calcium ionophores or other stimuli[16, 17], but the extent of physiological exosome secretion in non-immune cells is largely unknown.
What happens when exosomes reach an acceptor cell?
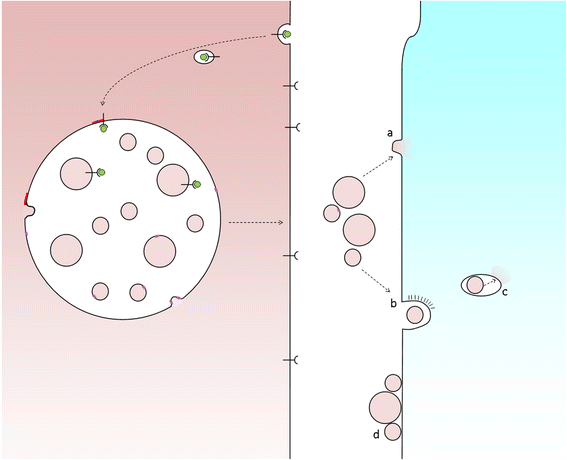
Exosome uptake by recipient cells. Fusion of MVBs with the cell surface releases ILVs as exosomes. In order for exosomes to elicit a response from recipient cells they might either fuse with plasma membrane (a) or be taken up whole via endocytosis (b), following which the exosome must be delivered to the cytosol, for example, via a back-fusion event (c). Alternatively, exosomes may attach to the surface of recipient cells to elicit a signalling response (d)
For intercellular transmission, various mechanisms of phagocytosis and endocytosis of extracellular vesicles have been described and which mechanism operates may depend upon vesicle size, which may in turn depend upon the cargo carried by the vesicle. In order for material to be released to an acceptor cell, exosomes must fuse with the host cell and this takes place via either direct fusion with the plasma membrane or a ‘back-fusion’ step from within a host endocytic organelle after the exosome has been engulfed. The process of back-fusion is not entirely clear, although it appears to require the unconventional lipid LBPA and protein Alix [19] (and is exploited by anthrax toxin lethal factor to escape from endosomes to the cytosol [20]).
Whether exosomes fuse with target cells or act via interactions with cell-surface proteins, or both, is another fundamental cell biology question that will need to be addressed if we are to understand the functions of exosomes.
So what are the consequences of all this information transfer? What biological functions have been established for exosomes?
Or, as with follicular dendritic cells, exosome-associated MHC II can be found on the surface of cell types that neither express MHC II nor secrete exosomes, indicating that exosomes are delivered from one cell type to another [18].
How exactly would exosomes from one cell influence the expression and activity of proteins in an acceptor cell?
So exosomes can also contribute to disease?
Of the neurodegenerative-associated proteins, only some are integral membrane proteins, that is, proteins inserted into lipid bilayers, rather than cytosolic. Sorting of proteins into ILVs (and thus exosomes) is easier to envisage for membrane proteins, where tags such as ubiquitin regulate where they end up. So far, the presence of both Aß [29] and PrPc [26] has in fact been shown in ILVs, though this has not been demonstrated for other membrane proteins, such as alpha-synuclein and tau.
The mechanism whereby cytosolic proteins may be sorted to ILVs/exosomes, however, is not clear. In order for cytosolic proteins to become concentrated in ILVs, they would require positive incorporation and sorting, possibly by membrane-associated components on endosomes. All we can say is that there is evidence that this does in fact happen; cytosolic factors such as miRNAs are enriched in exosomes relative to cytosol, indicating that sorting must occur whereby certain miRNAs are concentrated and others are not [30].
The means by which disease-associated factors spread between cells remains poorly understood and exosomes would provide a means for such transmission. The presence of exosomal proteins, such as Alix, in association with Alzheimer’s senile plaques strengthens the circumstantial case for exosomes as a mediator in such spread. The hope is that having a means to regulate exosome release and spread may be useful in combatting some of these diseases but much more basic biology needs to be established before then.
Now I’m confused—what determines what exosomes contain?
The mechanisms that concentrate cytosolic factors are currently unknown. Although it seems clear that miRNAs, for example, are enriched relative to the amount in their parent cells, and are not randomly incorporated into exosomes, it is not clear how some are enriched more than others. There are currently a few hypotheses for miRNA sorting, including sorting via sumoylated heterogeneous nuclear ribonucleoproteins [31] or by a miRNA-induced silencing complex (miRISC) [32].
Because of the difficulties in separating exosomes from other extracellular vesicles, it is likely that some cargos reported to be enriched in ‘exosomes’ may in fact be contained in contaminant vesicles that are not exosomes. While many researchers are very stringent about applying the labels ‘exosomes’ and ‘extracellular vesicles’ correctly, others unfortunately are not. In addition, as I have said before, cytosolic proteins are likely to be found in exosome preparations because the exosome lumen is made of cytosol.
So how exactly can you be sure that a given extracellular vesicle is an exosome and not something else?
One problem is that ILVs, and thus exosomes, represent an intermediate compartment of an intermediate. MVBs are not static organelles but rather undergo continuous maturation, in the course of which they gain and lose proteins. There will never be an exclusive marker for exosomes because any cargo on the ILV/exosome membrane must first be on the limiting membrane of the endosome and anything found inside must first come from the cytosol. A cargo may be concentrated on ILVs/exosomes but it will also be elsewhere. CD63 could be thought of as a pseudo-marker for exosomes. ILVs and exosomes are enriched in several such tetraspanins and my colleagues and I have show that CD63 is required for ESCRT-independent ILV formation [9]. Alix also appears to be concentrated in ILVs/exosomes [33], as does Tsg101, a component of ESCRT-I, which has been used as a marker of exosomes in numerous studies [33,34], although the presence of Tsg101 in ILVs or exosomes does not fit with conventional models of ILV formation. Although Tsg101 is involved in ESCRT-dependent ILV formation, as mentioned earlier, it, along with other ESCRT components, should disassociate from the endosomal membrane prior to an ILV pinching off the endosomal membrane to allow it to participate in further events [35]. Exactly when ESCRT-I components ‘fall off’ the membrane is unknown but it is conventionally thought to be prior to ILV formation, so Tsg101 should remain cytosolic and available for subsequent rounds of ILV formation. It is possible that some Tsg101 may be ‘swallowed’ into the forming ILV lumen, but levels should be negligible.
So are you saying there is no reliable marker for endosomes?
If they are so hard to characterize reliably, how are exosomes isolated and studied?
What would you say are the most important issues in exosome research?
References
-
Harding C, Heuser J, Stahl P. Receptor-mediated endocytosis of transferrin and recycling of the transferrin receptor in rat reticulocytes. J Cell Biol. 1983;97(2):329–39.
-
Pan BT, Teng K, Wu C, Adam M, Johnstone RM. Electron microscopic evidence for externalization of the transferrin receptor in vesicular form in sheep reticulocytes. J Cell Biol. 1985;101(3):942–8.
-
Johnstone RM, Adam M, Hammond JR, Orr L, Turbide C. Vesicle formation during reticulocyte maturation. Association of plasma membrane activities with released vesicles (exosomes). J Biol Chem. 1987;262(19):9412–20.
-
Raposo G, Nijman HW, Stoorvogel W, Liejendekker R, Harding CV, Melief CJ, et al. B lymphocytes secrete antigen-presenting vesicles. J Exp Med. 1996;183(3):1161–72.
-
Zitvogel L, Regnault A, Lozier A, Wolfers J, Flament C, Tenza D, et al. Eradication of established murine tumors using a novel cell-free vaccine: dendritic cell-derived exosomes. Nat Med. 1998;4(5):594–600.
-
Kim MS, Haney MJ, Zhao Y, Mahajan V, Deygen I, Klyachko NL, et al. Development of exosome-encapsulated paclitaxel to overcome MDR in cancer cells. Nanomedicine. 2015;12(3):655–64.
-
McDonald B, Martin-Serrano J. No strings attached: the ESCRT machinery in viral budding and cytokinesis. J Cell Sci. 2009;122(Pt 13):2167–77.
-
Stuffers S, Sem Wegner C, Stenmark H, Brech A. Multivesicular endosome biogenesis in the absence of ESCRTs. Traffic. 2009;10(7):925–37.
-
Edgar JR, Eden ER, Futter CE. Hrs- and CD63-dependent competing mechanisms make different sized endosomal intraluminal vesicles. Traffic. 2014;15(2):197–211.
-
Trajkovic K, Hsu C, Chiantia S, Rajendran L, Wenzel D, Wieland F, et al. Ceramide triggers budding of exosome vesicles into multivesicular endosomes. Science. 2008;319(5867):1244–7.
-
White IJ, Bailey LM, Aghakhani MR, Moss SE, Futter CE. EGF stimulates annexin 1-dependent inward vesiculation in a multivesicular endosome subpopulation. EMBO J. 2006;25(1):1–12.
-
Mobius W, Ohno-Iwashita Y, van Donselaar EG, Oorschot VM, Shimada Y, Fujimoto T, et al. Immunoelectron microscopic localization of cholesterol using biotinylated and non-cytolytic perfringolysin O. J Histochem Cytochem. 2002;50(1):43–55.
-
Buschow SI, Nolte-’t Hoen EN, van Niel G, Pols MS, ten Broeke T, Lauwen M, et al. MHC II in dendritic cells is targeted to lysosomes or T cell-induced exosomes via distinct multivesicular body pathways. Traffic. 2009;10(10):1528–42.
-
Blanchard N, Lankar D, Faure F, Regnault A, Dumont C, Raposo G, et al. TCR activation of human T cells induces the production of exosomes bearing the TCR/CD3/zeta complex. J Immunol. 2002;168(7):3235–41.
-
Muntasell A, Berger AC, Roche PA. T cell-induced secretion of MHC class II-peptide complexes on B cell exosomes. EMBO J. 2007;26(19):4263–72.
-
Savina A, Furlan M, Vidal M, Colombo MI. Exosome release is regulated by a calcium-dependent mechanism in K562 cells. J Biol Chem. 2003;278(22):20083–90.
-
Guo BB, Bellingham SA, Hill AF. Stimulating the release of exosomes increases the intercellular transfer of prions. J Biol Chem. 2016;291(10):5128–37.
-
Denzer K, van Eijk M, Kleijmeer MJ, Jakobson E, de Groot C, Geuze HJ. Follicular dendritic cells carry MHC class II-expressing microvesicles at their surface. J Immunol. 2000;165(3):1259–65.
-
Bissig C, Gruenberg J. ALIX and the multivesicular endosome: ALIX in Wonderland. Trends Cell Biol. 2014;24(1):19–25.
-
Abrami L, Lindsay M, Parton RG, Leppla SH, van der Goot FG. Membrane insertion of anthrax protective antigen and cytoplasmic delivery of lethal factor occur at different stages of the endocytic pathway. J Cell Biol. 2004;166(5):645–51.
-
Lo Cicero A, Delevoye C, Gilles-Marsens F, Loew D, Dingli F, Guere C, et al. Exosomes released by keratinocytes modulate melanocyte pigmentation. Nat Commun. 2015;6:7506.
-
Valadi H, Ekstrom K, Bossios A, Sjostrand M, Lee JJ, Lotvall JO. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol. 2007;9(6):654–9.
-
Melo SA, Sugimoto H, O’Connell JT, Kato N, Villanueva A, Vidal A, et al. Cancer exosomes perform cell-independent microRNA biogenesis and promote tumorigenesis. Cancer Cell. 2014;26(5):707–21.
-
Rajendran L, Honsho M, Zahn TR, Keller P, Geiger KD, Verkade P, et al. Alzheimer’s disease beta-amyloid peptides are released in association with exosomes. Proc Natl Acad Sci U S A. 2006;103(30):11172–7.
-
Saman S, Kim W, Raya M, Visnick Y, Miro S, Saman S, et al. Exosome-associated tau is secreted in tauopathy models and is selectively phosphorylated in cerebrospinal fluid in early Alzheimer disease. J Biol Chem. 2012;287(6):3842–9.
-
Fevrier B, Vilette D, Archer F, Loew D, Faigle W, Vidal M, et al. Cells release prions in association with exosomes. Proc Natl Acad Sci U S A. 2004;101(26):9683–8.
-
Emmanouilidou E, Melachroinou K, Roumeliotis T, Garbis SD, Ntzouni M, Margaritis LH, et al. Cell-produced alpha-synuclein is secreted in a calcium-dependent manner by exosomes and impacts neuronal survival. J Neurosci. 2010;30(20):6838–51.
-
Gomes C, Keller S, Altevogt P, Costa J. Evidence for secretion of Cu, Zn superoxide dismutase via exosomes from a cell model of amyotrophic lateral sclerosis. Neurosci Lett. 2007;428(1):43–6.
-
Guduric-Fuchs J, O’Connor A, Camp B, O’Neill CL, Medina RJ, Simpson DA. Selective extracellular vesicle-mediated export of an overlapping set of microRNAs from multiple cell types. BMC Genomics. 2012;13:357.
-
Edgar JR, Willen K, Gouras GK, Futter CE. ESCRTs regulate amyloid precursor protein sorting in multivesicular bodies and intracellular amyloid-beta accumulation. J Cell Sci. 2015;128(14):2520–8.
-
Villarroya-Beltri C, Gutierrez-Vazquez C, Sanchez-Cabo F, Perez-Hernandez D, Vazquez J, Martin-Cofreces N, et al. Sumoylated hnRNPA2B1 controls the sorting of miRNAs into exosomes through binding to specific motifs. Nat Commun. 2013;4:2980.
-
Fabian MR, Sonenberg N. The mechanics of miRNA-mediated gene silencing: a look under the hood of miRISC. Nat Struct Mol Biol. 2012;19(6):586–93.
-
Bobrie A, Colombo M, Krumeich S, Raposo G, Thery C. Diverse subpopulations of vesicles secreted by different intracellular mechanisms are present in exosome preparations obtained by differential ultracentrifugation. J Extracell Vesicles. 2012;1:18397.
-
Thery C, Boussac M, Veron P, Ricciardi-Castagnoli P, Raposo G, Garin J, et al. Proteomic analysis of dendritic cell-derived exosomes: a secreted subcellular compartment distinct from apoptotic vesicles. J Immunol. 2001;166(12):7309–18.
-
Hurley JH. The ESCRT complexes. Crit Rev Biochem Mol Biol. 2010;45(6):463–87.
-
ExoCarta. http://www.exocarta.org.
-
Thery C, Amigorena S, Raposo G, Clayton A. Isolation and characterization of exosomes from cell culture supernatants and biological fluids. Curr Protocols Cell Biol. 2006;Chapter 3:Unit 3 22.
-
Lotvall J, Hill AF, Hochberg F, Buzas EI, Di Vizio D, Gardiner C, et al. Minimal experimental requirements for definition of extracellular vesicles and their functions: a position statement from the International Society for Extracellular Vesicles. J Extracell Vesicles. 2014;3:26913.
Acknowledgements
Competing interests
The author declares that he has no competing interests.
- Published in Corporate News / Blog
Phase I/II Study of Safety and Preliminary Efficacy of Intravenous Allogeneic Mesenchymal Stem Cells in Chronic Stroke
Phase I/II Study of Safety and Preliminary Efficacy of Intravenous Allogeneic Mesenchymal Stem Cells in Chronic Stroke
Michael L. Levy, MD, PhD; John R. Crawford, MD; Nabil Dib, MD; Lev Verkh, PhD; Nikolai Tankovich, MD, PhD; Steven C. Cramer, MD
Background and Purpose—Stroke is a leading cause of long-term disability. Limited treatment options exist for patients with chronic stroke and substantial functional de?cits. The current study examined safety and preliminary ef?cacy estimates of intravenous allogeneic mesenchymal stem cells in this population.
Methods—Entry criteria included ischemic stroke >6 months prior and substantial impairment (National Institutes of Health Stroke Scale score =6) and disability. Enrollees received a single intravenous dose of allogeneic ischemia-tolerant mesenchymal stem cells. Phase 1 used a dose-escalation design (3 tiers, n=5 each). Phase 2 was an expanded safety cohort. The primary end point was safety over 1-year. Secondary end points examined behavioral change.
Results—In phase 1 (n=15), each dose (0.5, 1.0, and 1.5 million cells/kg body weight) was found safe, so phase 2 subjects (n=21) received 1.5 million cells/kg. At baseline, subjects (n=36) averaged 4.2±4.6 years poststroke, age 61.1±10.8 years, National Institutes of Health Stroke Scale score 8 (6.5–10), and Barthel Index 65±29. Two were lost to follow-up, one was withdrawn and 2 died (unrelated to study treatment). Of 15 serious adverse events, none was possibly or probably related to study treatment. Two mild adverse events were possibly related to study treatment, a urinary tract infection and intravenous site irritation. Treatment was safe based on serial exams, electrocardiograms, laboratory tests, and computed tomography scans of chest/abdomen/pelvis. All behavioral end points showed signi?cant gains over the 12-months of follow-up. For example, Barthel Index scores increased by 6.8±11.4 points (mean±SD) at 6-months (P=0.002) and by 10.8±15.5 points at 12-months (P<0.001) post-infusion; the proportion of patients achieving excellent functional outcome (Barthel score =95) increased from 11.4% at baseline to 27.3% at 6-months and to 35.5% at 12-months.
Conclusions—Intravenous transfusion of allogeneic ischemia-tolerant mesenchymal stem cell in patients with chronic stroke and substantial functional de?cits was safe and suggested behavioral gains. These data support proceeding to a randomized, placebo-controlled study of this therapy in this population.
Clinical Trial Registration—URL: http://www.clinicaltrials.gov. Unique identi?er: NCT01297413. (Stroke. 2019;50:00-00. DOI: 10.1161/STROKEAHA.119.026318.)
Key Words: abdomen ? brain ischemia ? neuroprotection ? pelvis ? reperfusion
Stroke is perennially among the leading causes of human disability1 and the leading neurological cause of lost dis-ability-adjusted life years.2 The mean survival after stroke is 6 to 7 years, and indeed more than 85% of patients live past the ?rst year poststroke,3 many with years of enduring disability. Many restorative therapies are under study to improve outcomes after stroke.4 Restorative therapies aim to improve patient outcomes by promoting the neural processes under-lying behavioral recovery,5 and are distinguished from acute therapies, such as reperfusion or neuroprotection, that aim to reduce initial injury. As such, restorative therapies often have a time window measured in days-months, or in some cases6–9 in years.
Mesenchymal stem cells (MSC), also known as mes-enchymal stromal cells, are among the leading restorative therapy candidates. Substantial preclinical data support the safety and ef?cacy of MSC as a restorative therapy to improve outcomes after stroke. For example, a meta-analysis reported that 44 of 46 preclinical stroke studies found MSC to be supe-rior to placebo,10 with effect sizes >1.0.
Initial human studies of MSC (or MSC-like cells) after stroke focused on autologous cell therapies,11–13 whereby bone marrow is taken from each patient to produce his/her own MSC batch, and found MSC infusion to be safe. MSC are rela-tively immunoprivileged given their very low levels of human leukocyte antigen molecule expression,14 a fact that opens the door to administration of allogeneic MSC. Allogeneic MSC Table 1. Entry Criteria have been found to be safe without use of concomitant im-munosuppression,15 and can be manufactured in a manner that enables broad clinical application. Studies of allogeneic MSC (or MSC-like cells) poststroke have focused on early time points (administration 24–48 hours poststroke)16 or used an invasive procedure to implant cells intracerebrally.17 Each approach has its relative advantages and disadvantages, and an intravenous method of introducing MSC if comparably ef-?cacious might facilitate widespread implementation and also avoid adverse events attributable to invasive procedures.
The current study was a phase I/II dose-escalation trial that examined effects of a single intravenous infusion of al-logeneic ischemia-tolerant MSC. The target population was patients with chronic ischemic stroke and substantial func-tional de?cits, a group for whom treatment options remain limited. The primary outcome was safety, based on serial measures of behavior, computed tomography (CT) scans, and laboratory testing. Preliminary estimates of treatment ef?cacy were also examined.
Table 1. Entry Criteria
| Inclusion criteria |
| 1. Age =18 y |
| 2. Ischemic stroke =6 mo prior, radiologically confirmed at initial diagnosis and at study enrolment |
| 3. Severe disability resulting from the index stroke, operationally defined as subject confined to a wheelchair or required to have home nursing care or needing assistance with activities of daily living |
| 4. No substantial improvement in neurological or functional status for the 2 mo before study enrolment |
| 5. NIHSS score 6–20 |
| 6. Life expectancy >12 mo |
| 7. Patient receiving standard of care secondary stroke prevention before enrolment |
| 8. Patient or a surrogate able to provide informed consent |
| 9. Reasonable expectation that the patient will receive standard posttreatment care and attend all scheduled study visits |
| 10. Adequate systemic organ function, specifically: Serum aspartate aminotransferase =2.5× upper limit of normal Serum alanine aminotransferase =2.5× upper limit of normal Total serum bilirubin =1.5× upper limit of normal Prothrombin time and partial thromboplastin time =1.25× upper limit of normal in subjects who are not receiving anti-thrombotic therapy Serum albumin =3.0 g/dL Absolute neutrophil count =1500/µL Platelet count =150 000/µL Hemoglobin =9.0 g/dL Serum creatinine =1.5× upper limit of normal Serum amylase or lipase =1.0× upper limit of normal |
| Exclusion criteria |
| 1. History of uncontrolled seizure disorder |
| 2. History of cancer within the past 5 y, with the exception of localized basal or squamous cell carcinoma |
| 3. History of cerebral neoplasm |
| 4. Positive for hepatitis B, C, or HIV |
| 5. Myocardial infarction within 6 months of study entry |
| 6. Presence of any other clinically significant medical or psychiatric condition, or laboratory abnormality, for which study participation would pose a safety risk in the judgment of the Investigator or Sponsor |
| 7. Findings on baseline computed tomography suggestive of subarachnoid or intracerebral hemorrhage within past 12 mo. |
| 8. Participation in another investigational drug or device study in the 3 mo before treatment |
| 9. History within the past year of drug or alcohol abuse |
| 10. Pregnant or lactating, or expectation to become pregnant during the study |
| 11. Allergy to bovine or porcine products |
NIHSS indicates National Institutes of Health Stroke Scale
Methods
Study Design
This was a phase I/II multi-center, open-label study that aimed to evaluate the safety and preliminary ef?cacy of a single intra-venous infusion of marrow-derived allogeneic ischemia-tolerant MSC. Entry criteria appear in Table 1 and in sum describe enroll-ment of adults with radiologically veri?ed chronic stable ischemic stroke and substantial impairment and functional de?cits. Patients were followed for one year after MSC infusion. The study made no restrictions on, and did not provide any forms of, medication or therapy (occupational, physical, or speech) during the follow-up year after infusion. All patients signed consent in accordance with local Institutional Review Board approval. This study was approved by the Food and DrugAdministration and was registered at clinical-trials.gov. The data that support the ?ndings of this study are avail-able from the corresponding author on reasonable request.
The study occurred in 2 parts, with part 1 being a dose-escala-tion study and part 2 being an expanded safety study based on part 1 ?ndings. Part 1 consisted of 3 cohorts (n=5 per cohort) enrolled sequentially in a dose-escalation manner, with subjects receiving one of 3 doses based on body weight, with a maximum dosage of 150 million cells. Cohort 1 received 0.5 million cells/kg of body weight; Cohort 2, 1.0 million cells/kg; and Cohort 3, 1.5 million cells/kg. The dose-escalation plan in part 1 required a review by the Data Safety Monitoring Board once the 5 subjects in Cohort 1 were treated and evaluated through study day 10. If safety was established, Cohort 2 was to proceed at the next highest dose, followed by a similar safety review before escalation to the highest dose in Cohort 3. Part 2 aimed to enroll an additional minimum of 20 subjects at the highest safe dose level determined in part 1. An additional interim review was conducted by the Data Safety Monitoring Board after the ?rst 5 patients were treated in part 2. Detailed stopping rules appear in the online-only Data Supplement (see Stopping Rules and Determination of Maximum Tolerated Dose).
The target dose of 1.5 million cells/kg corresponds to allometric scaling from animal studies. Our meta-analysis of preclinical studies of MSC after experimental ischemic stroke10 identi?ed 9 rodent stud-ies that transfused MSC using the intravenous route in the post-acute period. In each study, MSC provided substantial behavioral gains (effect sizes >1.0), using doses ranging from 3.6 to 12.4×106 MSC/kg body weight (mean dose of 10.1×106 MSC/kg). The approach to al-lometric scaling from animals to humans recommended by the Food and Drug Administration 18 uses a body surface area normalization, which for the mean value in rodents yields a comparable human dose of 1.6×106 MSC/kg.
Cell Manufacturing and Shipping
Manufacturing of MSC was performed at the GMP-compliant fa-cility of the sponsor, Stemedica Cell Technologies, Inc (San Diego, CA). MSC were grown from the bone marrow of a single human donor and are from the same batch used in prior preclinical19,20 and clinical21 studies. Cells were grown under low oxygen (5%) condi-tions. Such ischemia-tolerant MSC have advantages compared with those grown under normoxic conditions, for example, showing higher proliferation rate, expression of stem cell-related genes, production of key cytokines, and migration activity.21,22 Cells were harvested at passage 4 and expressed CD105, CD73, and CD90 surface markers, consistent with the International Society for Cellular Therapy de?-nition.23 Cells were cryopreserved by suspending in Cryostar CS10 freezing medium (BioLife Solutions, Bothell, WA) then stored in the vapor phase of liquid nitrogen. This parent cell bank was then tested for quality control including cell count, viability, appearance, and quantitative polymerase chain reaction for viruses including HIV, Epstein-Barr virus, cytomegalovirus, hepatitis B virus, parvovirus B19, and hepatitis C virus. Cryovials were shipped at =-150° C in a vapor phase liquid nitrogen shipper with temperature monitor.
Infusion of Investigational Product
Each site’s pharmacy prepared MSC for infusion per a study-pro-vided protocol. Cryovials (the number of which was based on the dose to be infused) were thawed and MSC were washed in, and then suspended in, Lactated Ringer’s solution at a concentration of 1×106 cells/mL using one to three 60 mL syringes. The suspension then un-derwent ?nal testing before being released for intravenous infusion, consisting of cell count, endotoxin, Gram stain, and review of appear-ance. Cell count was performed using 0.1% Trypan Blue and a hema-cytometer, which also yielded % cell viability. The minimum percent cell viability was required to be =70% for the cells to be released. A sample was also sent for subsequent sterility testing. After release by the pharmacy, the ?nal formulation was stored at 2° to 8°C and infused within 8 hours of preparation.
MSC Administration
Before MSC infusion, a 0.1 mL aliquot of the ?nal MSC formulation was injected intradermally; any subject showing a positive reaction (eg, wheal with erythema) would not be infused. Cells were admin-istered intravenously via metered-dose syringe pump at 2 mL/min. Patients remained in the inpatient telemetry unit for observation until clinically stable.
Patient Assessments
Patients had frequent monitoring until discharged from the telem-etry unit. After discharge, patients had safety evaluations on day 2, 3, 4, and 10, then again on month, 1, 3, 6, 9, and 12. Adverse events were coded according to the MedDRA adverse event dic-tionary. The relationship that adverse events had to the investiga-tional product was assessed by the site investigator. Patients were followed for one year using tests of behavior, serology, blood chem-istry and cell counts, electrocardiogram, urine, and CT of chest, abdomen, and pelvis. The full schedule of assessments appears in Table SI in the online-only Data Supplement.
Statistics
The primary study end point was safety and tolerability, evaluated in all subjects who received any portion of an infusion, and determined by the incidence/severity of adverse events, clinically signi?cant changes on laboratory and imaging tests, vital signs, and physical plus neurological examinations. Four secondary end points were scored serially to derive preliminary estimates of ef?cacy: National Institutes of Health Stroke Scale, Barthel Index (BI), Mini-Mental Status Exam, and Geriatric Depression Scale. For each, the change from baseline was evaluated using Wilcoxon signed-rank test, with primary analysis of preliminary ef?cacy being change from baseline to 6 months post-infusion, and analysis including all subjects who re-ceived an infusion except for one subject who failed to return after the day 10 visit for all visits (except for month 9 follow-up). For any sub-ject missing 6-month data, 9-month or 12-month data were substituted for this analysis, otherwise missing data were not imputed. Data were analyzed using R statistical software. Given the exploratory nature of this study, sample size was selected as appropriate for detection of any safety concerns in an early phase clinical trial.
Results
Subjects
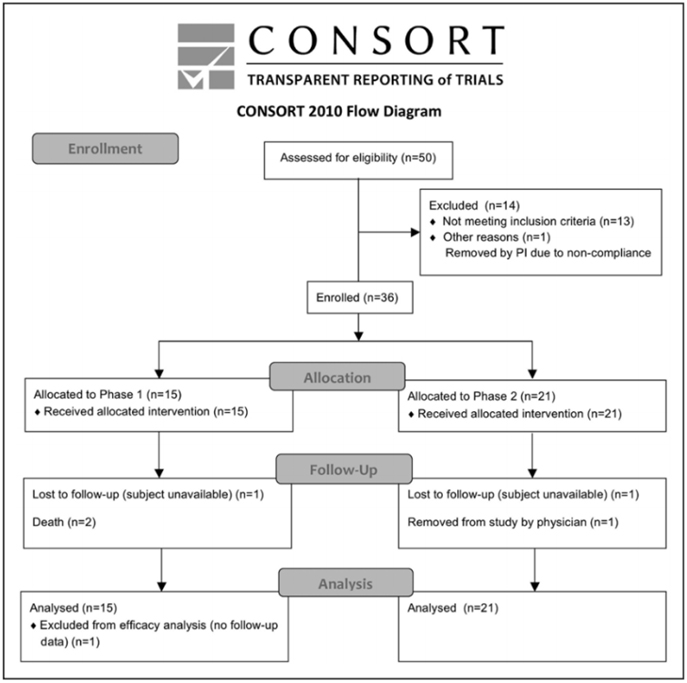
Of 50 subjects who seemed eligible on prescreening, 36 were enrolled and received treatment from March 14, 2011 to December 15, 2016 (Figure and Table 2). There were 13 subjects enrolled at the University of California, San Diego, 19 subjects at Arizona, and 4 subjects at the University of California, Irvine. Interim safety reviews disclosed no con-cerns, and so 5 subjects received 0.5×106 cells/kg in part 1/ Cohort 1, 5 subjects received 1.0×106 cells/kg in part 1/Cohort 2, 5 subjects received 1.5×106 cells/kg in part 1/Cohort 3, and all 21 subjects in part 2 received 1.5×106 cells/kg. For the 15 subjects in part 1, 12 completed the study, 2 died of unrelated causes (coronary artery disease 6 months post-infusion and sepsis 1 month after infusion), and 1 was lost to follow-up after day 10 (reappearing only for the month 9 follow-up visit). For the 21 subjects in part 2, 19 completed the study, 1 was lost to follow-up after month 6, and 1 was withdrawn by the site PI after month 6 due to treatment with another in-vestigational product. Of the 36 subjects enrolled, the planned dose was delivered within 2 mL (ie, within 2×106 cells) of the target in 26 subjects, whereas in 10 subjects a median of 7.6 (interquartile range, 4.4–10.25) mL (ie, 7.6×106 cells) was not infused as planned, which represented a median of 6.5% (5.3– 9.8) of the intended dose. A total of 179 protocol deviations were reported, mainly related to scheduling study visits or study testing (Table SII in the online-only Data Supplement).
Figure. CONSORT diagram.
Table 2. Baseline Subject Characteristics
| Part1 | Part 2 | Total | |||
| Cohort 1 | Cohort 2 | Cohort 3 | |||
| n | 5 | 5 | 5 | 21 | 36 |
| Sex | |||||
| Male | 5 (100%) | 4 (80%) | 4 (80%) | 14 (66.67%) | 27 (75%) |
| Female | 0 (0%) | 1 (20%) | 1 (20%) | 7 (33.33%) | 9 (25%) |
| Age, y | 50.8:±9.8 [40—62] | 56.8±11.1 [39—69] | 68.8±11.58 [53—84] | 62.8±9.2 [51—83] | 61.1±10.8 [39—84] |
| Race | |||||
| White | 4 (80%) | 3 (60%) | 5 (100%) | 17 (80.95%) | 29 (80.56%) |
| Asian | 0 (0%) | 1 (20%) | 0 (0%) | 0 (0%) | 1 (2.78%) |
| American Indian/Alaskan Native | 0 (0%) | 1 (20%) | 0 (0%) | 0 (0%) | 1 (2.78%) |
| Native Hawaiian/Pacific Islander | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) |
| Black | 1 (20%) | 0 (0%) | 0 (0%) | 1 (4.76%) | 2 (5.56%) |
| Other | 0 (0%) | 0 (0%) | 0 (0%) | 3 (14.29%) | 3 (8.33%) |
| Ethnicity | |||||
| Hispanic or Latino | 0 (0%) | 0 (0%) | 0 (0%) | 2 (9.52%) | 2 (5.56%) |
| Non-Hispanic or Non-Latino | 5 (100%) | 5 (100%) | 5 (100%) | 19 (90.48%) | 34 (94.44%) |
| Living situation | |||||
| At home | 5 (100%) | 5 (100%) | 3 (60%) | 19 (90.48%) | 32 (88.89%) |
| In a living facility | 0 (0%) | 0 (0%) | 2 (40%) | 2 (9.52%) | 4 (11.11%) |
| Time from stroke to infusion, y | 1.6±0.9 [0.6—2.9] | 7.7±5.0 [1.1—14.5] | 4.1±2.2 [1.7—7.0] | 4.0±5.0 [0.7—24.8] | 4.2±4.6 [0.6—24.8] |
Values are counts (%) else mean±SD. Values in brackets indicate range.
Safety
A total of 15 serious adverse events were reported.These were wide-ranging in nature, for example, infections, vascular dis-orders, and pain syndromes (for full details, see Table SIII in the online-only Data Supplement). All serious adverse events were deemed unrelated or unlikely related to the investiga-tional product. A total of 109 adverse events were reported, of which 2, both mild, were considered by the site investigator to be possibly related to the investigational product: one uri-nary tract infection and one report of intravenous site irrita-tion. Both adverse events recovered completely.
Study testing disclosed no safety concerns. No subject showed a preinfusion positive reaction to intradermal testing. Serial physical exams and blood testing did not disclose any signi?cant ?ndings. Only one of the serial electrocardiograms was thought to have clinically signi?cant ?ndings, in a subject with moderate intraventricular conduction delay, only at the 1-month follow-up visit. Similarly, across serial CT scans of the chest, abdomen, and pelvis, only one was considered clinically signi?cant, a soft tissue density in the anterior abdominal wall seen at 6-months that was stable when reimaged at 12-months.
Behavioral Effects
Across all subjects, improvements were seen in National Institutes of Health Stroke Scale, BI, Mini-Mental Status Exam, and Geriatric Depression Scale scores at both the 6-month and the 12-month follow-up visits (Table 3). These were statistically signi?cant, generally stable over time, and clinically modest in magnitude. Most ?ndings would sur-vive correction for multiple comparisons. Changes in the BI suggest clinical utility, with a 6.8 point gain by 6-months that grew to a 10.8 point gain by 12-months post-infusion (P<0.001), and with the proportion of patients achieving ex-cellent functional outcome (Barthel score =95) increasing from 11.4% (4/35) at baseline to 9/33 (27.3%) at 6-months to 35.5% (11/31) at 12-months.
Discussion
Stroke is a major cause of human disability. This can be reduced by acute therapies that are introduced in the early hours poststroke to reduce initial injury, and by restorative therapies that are introduced days, months, or years poststroke to promote neural repair. Allogeneic MSC show substantial favorable effects in preclinical studies, including when intro-duced via the intravenous route.10 The current study found a single intravenous infusion of allogeneic MSC to be safe and potentially associated with functional improvement.
The current study is the largest trial of intravenous MSC in patients with chronic stroke and the ?rst to evaluate alloge-neic MSC therapy in this population. It is also the ?rst human stroke study to evaluate MSC grown under hypoxic conditions, which favorably affects cell proliferation, gene expression, cy-tokine production, and migration.21,22 Intravenous infusion of MSCwasfoundtobesafein36patientswhohadchronicstroke with substantial functional de?cits. Across 3 escalating doses, treatment-related adverse events were infrequent, mild, and transient. Serial assessments of exam, laboratory testing, elec-tro cardiogram, and CT scans of chest/abdomen/pelvis disclosed no safety concerns, with limited subject dropout. These results are consistent with the overall excellent safety record that MSC have in clinical trials of human subjects across numerous non-cerebrovascular diagnoses15,24–27 and in stroke trials.11–13,16,17,28,29
Patients with stroke in the chronic stage generally show functional decline; however, enrollees in the current study showed 12 months of continued functional improvement. In general, recovery from stroke-related de?cits shows a bimodal time course. Initially, most stroke survivors show some degree of spontaneous recovery, for example, during the initial months for the motor system.30 Within a year of stroke onset, however, a signi?cant decline in function is commonly seen.31–34 This is signi?cant given that few treat-ment options are available to improve function in patients in the chronic phase of stroke. In the current study, behav-ioral gains were seen, though were modest in magnitude. However, a 2-point improvement in the National Institutes of Health Stroke Scale score (Table 3) in the setting of chronic stroke, if veri?ed in a larger controlled study, might be regarded as important. Also, the mean gain in BI from base-line grew to 10.8 points by 12 month-poststroke (P<0.001), higher than the BI minimal clinically important difference of 9.25 points.35 Furthermore, the proportion of patients with an excellent functional outcome (BI score =95) increased from 11.4% at baseline to 27.3% at 6-months and to 35.5% at 12-months (Table 3). This 12-month period of continued functional improvement is consistent with preclinical studies examining the distribution of systemically administered MSC: intravenous MSC given early after stroke initially localize to lungs then spleen, then increase within the region of brain ischemia,36 and by 30 days poststroke are concen-trated in the peri-infarct region.37 At one year, most surviving MSC are in the peri-infarct region, with very few present in other organs.38 Patients also showed signi?cant improvement in the Mini-Mental Status Exam and Geriatric Depression Scale (Table 3), changes that were largely sustained at 12 months post-infusion, suggesting that MSC have broad effects on brain function. These ?ndings require veri?cation in a larger, controlled study but raise hope that this interven-tion could improve functional status in the chronic stroke setting. Future studies might also incorporate modality-spe-ci?c outcome measures30 to provide more granular assessments of behavioral gains in individual neural systems.
Table 3. Behavioral Change Over Time
| Mini-Mental Status Exam score | n | P Value | |
| Baseline | 24.2±6.0 | 35 | |
| Change to 6 mo | 1.8±2.8 | 32 | <0.001 |
| Change to 12 mo | 1.3±2.7 | 31 | 0.017 |
| NIHSS score | |||
| Baseline | 8 [6.5 to 10] | 35 | |
| Change to 6 mo | -1 [-2.25 to 0] | 32 | <0.001 |
| Change to 12 mo | -2 [-3.5 to -0.5] | 21 | <0.001 |
| Geriatric depression scale score | |||
| Baseline | 5.1±3.5 | 35 | |
| Change to 6 mo | -1.6±3.8 | 32 | 0.015 |
| Change to 12 mo | -1.4±3.8 | 31 | |
| Barthel Index (score) | |||
| Baseline | 65±28.7 | 35 | |
| Change to 6 mo | 6.8±11.4 | 33 | 0.002 |
| Change to 12 mo | 10.8±15.5 | 31 | <0.001 |
| Barthel Index (% =95) | |||
| Proportion at baseline | 4 (11.4%) | 35 | |
| Proportion at 6 mo | 9 (27.3%) | 33 | 0.015 |
| Proportion at 12 mo | 11 (35.5%) | 31 | 0.01 |
Values are mean±SD or median (interquartile range) across all enrollees. Specific data for part 1 and part 2 appear in Table SIV in the online-only Data Supplement. NIHSS indicates National Institutes of Health Stroke Scale.
Meta-analysis of MSC effects in animals with experimental ischemic stroke10 showed large effect sizes that remained sub-stantial after adjusting for potential publication bias and was robust across species, delivery route, time of administration in relation to stroke, and dose. The longest time period when MSC have been introduced poststroke in preclinical studies is 1 month39 or 4 to 6 weeks40 post-infarct. The current ?ndings in patients who were many months poststroke (Table 2) sug-gest the need for bidirectional translation, that is, translation of bedside experience to inform preclinical studies.41,42
There are several strengths to this study. Enrollees had substantial functional de?cits in the chronic stage of stroke, a population that numbers in the millions, for whom treatment options remain limited. The infused cells were allogeneic, an approach made possible by the relatively immunoprivileged nature of MSC,14 which eliminates the need for immuno-suppression15 and which, as compared with autologous cell therapies, enables treatment protocols that can be broadly implemented in the stroke population. A dose-escalation study design was used to evaluate safety. Cell culture was limited to 4 passages, a potential advantage given that higher number of passages (and thus cell divisions) adversely affect MSC features such as proliferation, differentiation, homing, and viability.43–45 Safety was assessed across multiple modalities, including chest/abdomen/pelvis CT and extensive laboratory testing, for a 1-year period.
There are also important weaknesses. As this study was focused on safety, no control group was included, which complicates interpretation of observed behavioral gains (Table 3). Mechanism of action was not studied. Cell thera-pies improving outcomes in the chronic phase likely act via multiple mechanisms that include release of growth factors and anti-in?ammatory effects, and possibly exosomes,46,47 which can be evaluated in subsequent trials. Restorative ther-apies after stroke often provide maximal bene?t when paired with appropriate training,48 but this was not provided in the current safety study.
The current study demonstrated safety of intravenous MSC in patients with chronic stroke who had substantial functional de?cits. Results also suggest functional bene?t, although this requires veri?cation in a controlled study. Together, these ?ndings support further study of intravenous allogeneic MSC in patients with chronic stroke, including evaluation of mech-anism of action.
Sources of Funding
This study was supported by Stemedica Cell Technologies, Inc.
Disclosures
Dr Levy is on the Scienti?c Advisory Board for KOH Robotics. Dr Dib has served as a consultant for J&J Consulting and ISCTR Consulting. Dr Verkh is Chief Regulatory and Clinical Development Of?cer at Stemedica Cell Technologies, Inc. Dr Tankovich is the President and Chief Medical Of?cer for Stemedica Cell Technologies, Inc. Dr Cramer has served as a consultant for Abbvie, Constant Therapeutics, MicroTransponder, Neurolutions, Regenera, SanBio, Stemedica, Biogen, Fuji?lm Toyama Chemical, and TRCare. The other authors report no con?icts.
References
- Feigin VL, Lawes CM, Bennett DA, Anderson CS. Stroke epidemiology: a review of population-based studies of incidence, prevalence, and case-fatality in the late 20th century. Lancet Neurol. 2003;2:43–53.
- Johnston SC, Hauser SL. Neurological disease on the global agenda. Ann Neurol. 2008;64:A11–A12.
- Lloyd-Jones D, Adams RJ, Brown TM, Carnethon M, Dai S, De Simone G, et al. Heart disease and stroke statistics–2010 update: a report from the American Heart Association. Circulation. 2010;121:e46–e215.
- Lin DJ, Finklestein SP, Cramer SC. New directions in treat-ments targeting stroke recovery. Stroke. 2018;49:3107–3114. doi: 10.1161/STROKEAHA.118.021359
- Cramer SC. Repairing the human brain after stroke. II. Restorative thera-pies. Ann Neurol. 2008;63:549–560. doi: 10.1002/ana.21412
- Wolf SL, Winstein CJ, Miller JP, Taub E, Uswatte G, Morris D, et al; EXCITE Investigators. Effect of constraint-induced movement therapy on upper extremity function 3 to 9 months after stroke: the EXCITE randomized clinical trial. JAMA. 2006;296:2095–2104. doi: 10.1001/jama.296.17.2095
- McCabe J, Monkiewicz M, Holcomb J, Pundik S, Daly JJ. Comparison of robotics, functional electrical stimulation, and motor learning methods for treatment of persistent upper extremity dysfunction after stroke: a randomized controlled trial. Arch Phys Med Rehabil. 2015;96:981–990. doi: 10.1016/j.apmr.2014.10.022
- Dodakian L, McKenzie AL, Le V, See J, Pearson-Fuhrhop K, Burke Quinlan E, et al. A home-based telerehabilitation program for patients with stroke. Neurorehabil Neural Repair. 2017;31:923–933. doi: 10.1177/1545968317733818
- Ward NS, Brander F, Kelly K. Intensive upper limb neurorehabilita-tion in chronic stroke: outcomes from the Queen Square programme. J Neurol Neurosurg Psychiatry. 2019;90:498–506. doi: 10.1136/jnnp-2018-319954
- Vu Q, Xie K, Eckert M, Zhao W, Cramer SC. Meta-analysis of preclinical studies of mesenchymal stromal cells for ischemic stroke. Neurology. 2014;82:1277–1286. doi: 10.1212/WNL.0000000000000278
- Bang OY, Lee JS, Lee PH, Lee G. Autologous mesenchymal stem cell transplantation in stroke patients. Ann Neurol. 2005;57:874–882. doi: 10.1002/ana.20501
- Honmou O, Houkin K, Matsunaga T, Niitsu Y, Ishiai S, Onodera R, et al. Intravenous administration of auto serum-expanded autologous mes-enchymal stem cells in stroke. Brain. 2011;134(pt 6):1790–1807. doi: 10.1093/brain/awr063
- Bhasin A, Srivastava MV, Kumaran SS, Mohanty S, Bhatia R, Bose S, et al. Autologous mesenchymal stem cells in chronic stroke. Cerebrovasc Dis Extra. 2011;1:93–104. doi: 10.1159/000333381
- Le Blanc K, Tammik C, Rosendahl K, Zetterberg E, Ringdén O. HLA expression and immunologic properties of differentiated and undifferen-tiated mesenchymal stem cells. Exp Hematol. 2003;31:890–896.
- Lalu MM, McIntyre L, Pugliese C, Fergusson D, Winston BW, Marshall JC, et al; Canadian Critical Care Trials Group. Safety of cell therapy with mesenchymal stromal cells (SafeCell): a systematic re-view and meta-analysis of clinical trials. PLoS One. 2012;7:e47559. doi: 10.1371/journal.pone.0047559
- Hess DC, Wechsler LR, Clark WM, Savitz SI, Ford GA, Chiu D, et al. Safety and ef?cacy of multipotent adult progenitor cells in acute ischaemic stroke (MASTERS): a randomised, double-blind, pla-cebo-controlled, phase 2 trial. Lancet Neurol. 2017;16:360–368. doi: 10.1016/S1474-4422(17)30046-7
- Steinberg GK, Kondziolka D, Wechsler LR, Lunsford LD, Coburn ML, Billigen JB, et al. Clinical outcomes of transplanted modi?ed bone marrow-derived mesenchymal stem cells in stroke: a phase 1/2a study. Stroke. 2016;47:1817–1824. doi: 10.1161/STROKEAHA.116.012995
- Guidance for Industry: Estimating the Maximum Safe Starting Dose in Initial Clinical Trials for Therapeutics in Adult Healthy Volunteers. 2005. www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatory Information/Guidances/ucm078932.pdf.Accessed June 22, 2019.
- Luger D, Lipinski MJ, Westman PC, Glover DK, Dimastromatteo J, Frias JC, et al. Intravenously delivered mesenchymal stem cells: sys-temic anti-in?ammatory effects improve left ventricular dysfunction in acute myocardial infarction and ischemic cardiomyopathy. Circ Res. 2017;120:1598–1613. doi: 10.1161/CIRCRESAHA.117.310599
- Harach T, Jammes F, Muller C, Duthilleul N, Cheatham V, Zufferey V, et al. Administrations of human adult ischemia-tolerant mesenchymal stem cells and factors reduce amyloid beta pathology in a mouse model of Alzheimer ’s disease. Neurobiol Aging. 2017;51:83–96. doi: 10.1016/j.neurobiolaging.2016.11.009
- Butler J, Epstein SE, Greene SJ, Quyyumi AA, Sikora S, Kim RJ, et al. Intravenous allogeneic mesenchymal stem cells for nonischemic car-diomyopathy: safety and ef?cacy results of a phase II-a randomized trial. Circ Res. 2017;120:332–340. doi: 10.1161/CIRCRESAHA. 116.309717
- Vertelov G, Kharazi L, Muralidhar MG, Sanati G, Tankovich T, Kharazi A. High targeted migration of human mesenchymal stem cells grown in hypoxia is associated with enhanced activation of RhoA. Stem Cell Res Ther. 2013;4:5. doi: 10.1186/scrt153
- Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, et al. Minimal criteria for de?ning multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position state-ment. Cytotherapy. 2006;8:315–317. doi: 10.1080/14653240600855905
- Devine SM. Mesenchymal stem cells: will they have a role in the clinic? J Cell Biochem Suppl. 2002;38:73–79.
- Hil?ker A, Kasper C, Hass R, Haverich A. Mesenchymal stem cells and progenitor cells in connective tissue engineering and regenerative medicine: is there a future for transplantation? Langenbecks Arch Surg. 2011;396:489–497. doi: 10.1007/s00423-011-0762-2
- Williams AR, Hare JM. Mesenchymal stem cells: biology, pathophys-iology, translational ?ndings, and therapeutic implications for cardiac disease. Circ Res. 2011;109:923–940. doi: 10.1161/CIRCRESAHA. 111.243147
- Figueroa FE, Carrión F, Villanueva S, Khoury M. Mesenchymal stem cell treatment for autoimmune diseases: a critical review. Biol Res. 2012;45:269–277. doi: 10.4067/S0716-97602012000300008
- Tsang KS, Ng CPS, Zhu XL, Wong GKC, Lu G, Ahuja AT, et al. Phase I/II randomized controlled trial of autologous bone marrow-derived mesenchymal stem cell therapy for chronic stroke. World J Stem Cells. 2017;9:133–143. doi: 10.4252/wjsc.v9.i8.133
- Savitz SI,Yavagal D, Rappard G, Likosky W, Rutledge N, Graffagnino C, et al.A phase 2 randomized, sham-controlled trial of internal carotid artery infusion of autologous bone marrow-derived ALD-401 cells in patients with recent stable ischemic stroke (RECOVER-Stroke). Circulation. 2019;139:192–205. doi: 10.1161/CIRCULATIONAHA.117.030659
- Cramer SC, Koroshetz WJ, Finklestein SP. The case for modality-speci?c outcome measures in clinical trials of stroke recovery-pro-moting agents. Stroke. 2007;38:1393–1395. doi: 10.1161/01.STR. 0000260087.67462.80
- van de Port IG, Kwakkel G, van Wijk I, Lindeman E. Susceptibility to deterioration of mobility long-term after stroke: a prospective cohort study. Stroke. 2006;37:167–171.
- Dhamoon MS, Moon YP, Paik MC, Boden-Albala B, Rundek T, Sacco RL, et al. Long-term functional recovery after ?rst ischemic stroke: the Northern Manhattan Study. Stroke. 2009;40:2805–2811. doi: 10.1161/STROKEAHA.109.549576
- Wondergem R, Pisters MF, Wouters EJ, Olthof N, de Bie RA, Visser-Meily JM, et al. The course of activities in daily living: who is at risk for decline after ?rst ever stroke? Cerebrovasc Dis. 2017;43:1–8. doi: 10.1159/000451034
- Sennfalt S, Norrving B, Petersson J, Ullberg T. Long-term sur-vival and function after stroke. Stroke. 2019;50:53–61. doi: 10.1161/STROKEAHA.118.022913
- Hsieh YW, Wang CH, Wu SC, Chen PC, Sheu CF, Hsieh CL. Establishing the minimal clinically important difference of the Barthel Index in stroke patients. Neurorehabil Neural Repair. 2007;21:233– 238. doi: 10.1177/1545968306294729
- Detante O, Moisan A, Dimastromatteo J, Richard MJ, Riou L, Grillon E, et al. Intravenous administration of 99mTc-HMPAO-labeled human mes-enchymal stem cells after stroke: in vivo imaging and biodistribution. Cell Transplant. 2009;18:1369–1379. doi: 10.3727/096368909X474230
- Sykova E, Jendelova P. In vivo tracking of stem cells in brain and spinal cord injury. Prog Brain Res. 2007;161:367–383. doi: 10.1016/S0079-6123(06)61026-1
- Shen LH, Li Y, Chen J, Cui Y, Zhang C, Kapke A, et al. One-year follow-up after bone marrow stromal cell treatment in mid-dle-aged female rats with stroke. Stroke. 2007;38:2150–2156. doi: 10.1161/STROKEAHA.106.481218
- Shen LH, Li Y, Chen J, Zacharek A, Gao Q, Kapke A, et al. Therapeutic bene?t of bone marrow stromal cells administered 1 month after stroke. J Cereb Blood Flow Metab. 2007;27:6–13. doi: 10.1038/sj.jcbfm.9600311
- Yasuhara T, Matsukawa N, Hara K, Maki M, Ali MM, Yu SJ, et al. Notch-induced rat and human bone marrow stromal cell grafts reduce ischemic cell loss and ameliorate behavioral de?cits in chronic stroke an-imals. Stem Cells Dev. 2009;18:1501–1514. doi: 10.1089/scd.2009.0011
- Bosetti F, Koenig JI, Ayata C, Back SA, Becker K, Broderick JP, et al. Translational stroke research: vision and opportunities. Stroke. 2017;48:2632–2637. doi: 10.1161/STROKEAHA.117.017112
- Armstead WM, Vavilala MS. Translational approach towards deter-mining the role of cerebral autoregulation in outcome after traumatic brain injury. Exp Neurol. 2019;317:291–297. doi: 10.1016/j.expneurol. 2019.03.015
- Baxter MA, Wynn RF, Jowitt SN, Wraith JE, Fairbairn LJ, Bellantuono I. Study of telomere length reveals rapid aging of human marrow stromal cells following in vitro expansion. Stem Cells. 2004;22:675– 682. doi: 10.1634/stemcells.22-5-675
- Izadpanah R, Kaushal D, Kriedt C, Tsien F, Patel B, Dufour J, et al. Long-term in vitro expansion alters the biology of adult mesenchymal stem cells. Cancer Res. 2008;68:4229–4238. doi: 10.1158/0008-5472. CAN-07-5272
- Galipeau J. The mesenchymal stromal cells dilemma–does a negative phase III trial of random donor mesenchymal stromal cells in steroid-resistant graft-versus-host disease represent a death knell or a bump in the road? Cytotherapy. 2013;15:2–8. doi: 10.1016/j.jcyt.2012.10.002
- Wechsler LR, Bates D, Stroemer P, Andrews-Zwilling YS, Aizman I. Cell therapy for chronic stroke. Stroke. 2018;49:1066–1074. doi: 10.1161/STROKEAHA.117.018290
- Chen J, Chopp M. Exosome therapy for stroke. Stroke. 2018;49:1083– 1090. doi: 10.1161/STROKEAHA.117.018292
- Cramer SC, Sur M, Dobkin BH, O’Brien C, Sanger TD, Trojanowski JQ, et al. Harnessing neuroplasticity for clinical applications. Brain. 2011;134(pt 6):1591–1609. doi: 10.1093/brain/awr039
- Published in Corporate News / Blog
Biodistribution, migration and homing of systemically applied mesenchymal stem/stromal cells
Biodistribution, migration and homing of systemically applied mesenchymal stem/stromal cells
Abstract
Mesenchymal stem/stromal cells (MSCs) are increasingly used as an intravenously applied cellular therapeutic. They were found to be potent in situations such as tissue repair or severe inflammation. Still, data are lacking with regard to the biodistribution of MSCs, their cellular or molecular target structures, and the mechanisms by which MSCs reach these targets. This review discusses current hypotheses for how MSCs can reach tissue sites. Both preclinical and clinical studies using MSCs applied intravenously or intra-arterially are discussed in the context of our current understanding of how MSCs might work in physiological and pathological situations.
Background
In the 1970s, Friedenstein and colleagues [1] first reported that locally applied culture-expanded populations of bone marrow stroma-derived fibroblastic cells remained at their injection sites under the kidney capsule, where an ectopic hematopoiesis was initiated. Later, Arnold Caplan’s group described mesenchymal stem/stromal cells (MSCs) as multipotent mesenchymal cell populations which can differentiate into several tissue types, and demonstrated roles for MSCs in the regeneration of bone, cartilage or ligaments in animal and clinical studies [2–4]. In these studies, however, transplanted cells were followed, if at all, at the site of transplantation, and biodistribution was not an issue.
By the year 2000, clinicians had become increasingly interested in intravenously applied MSCs. Pivotal studies by the group of Horwitz in children with osteogenesis imperfecta, an inherited enzyme deficiency of collagen synthesis by mesenchymal cells in bone, opened the field for intravenous use of MSCs. This concept started from the observation that bone marrow transplantation can provide stromal cells able to synthesize intact collagen type I, replacing deficient patient cell function and ameliorating disease symptoms [5]. Therefore, the authors concluded that transplantation of isolated healthy allogeneic MSCs might cure the disease. This implies homing of transplanted MSCs to sites in bone marrow and/or bone. Efficacy was noted in all six infants treated [5]. Children who received transplants showed improved growth rates and started to synthesize intact bone. Engraftment of donor-type MSC-derived osteoblasts was shown using bone specimens and microsatellite DNA marker analysis. In a second study [6], these authors showed that autologous, enzyme-deficient MSCs transduced with a copy of the intact gene resulted in normal collagen production in bone cavities. Moreover, children who received transplants approached growth curves similar to the children transplanted with allogeneic complete bone marrow [6]. This pioneering work provided the basis for the successful application of MSCs using the intravenous route in other clinical entities.
Establishment of methods to track intravenously administered MSCs
After 2000, the therapeutic use of MSCs by intravenous administration was explored by a number of studies in animals and also humans. These studies used various ways to label culture-expanded MSCs, and to track them in different tissues over time. The tissue source of the MSCs was in most cases not decisive, and cells from various tissue sources were explored. The labeling methodologies used included radioactive labeling of MSCs, labeling with fluorescent vital dyes, contrast agents, transduction with reporter genes, or the use of donor cell-specific DNA markers such as microsatellites [7–11] (reviewed in [12]). The labeling methodologies were, in part, designed to detect only short-term homing of MSCs. In addition, they do not enable the determination of whether detected cells are still alive. These studies were mainly conducted in rodents and nonhuman primates and mostly in non-injury situations. The main common results of these studies were that: MSCs distribute to a variety of tissues after intravenous (i.v.) injection; MSCs are detectable at low or very low frequencies in tissues after transplantation; and signals from the injected cells were found early after administration of the MSCs at the highest frequencies in the lungs, followed by liver and spleen.
The observed biodistribution patterns were confirmed by studies in humans. In patients with mammary carcinoma, Koç et al. [13] demonstrated that i.v. MSCs were well-tolerated in patients at a dose of one million MSCs/kg body weight; however, the cells were trackable in blood only. The data were confirmed in patients with liver cirrhosis using 111In-oxine labeled MSCs, which were found to first accumulate in the lungs followed by continuous increases in liver and spleen up to day 10 after administration [14]. The proportion of accumulation in lung decreased from about 35 % early after transplantation to 2 % or less by day 10, whereas spleen had the highest signals by day 10 after transplant. These results confirm a similar overt biodistribution of MSCs in lung, liver and spleen in humans to that observed in animal models.
Expression of cell adhesion molecules by MSCs as a basis for their interaction with endothelial cells and tissue-directed extravasation
In theory, the main prerequisite for the interaction of transplanted MSCs with endothelial cells are adhesion molecules present on the cell surface of MSCs, and expression of appropriate adhesion counter-receptors on endothelial cells. MSCs (most investigations were performed in human MSCs (hMSCs)) have shown deficits in receptor binding to selectins and/or their ligands. They lack expression of L-selectin, and their E-selectin ligand (CD44) is not functional [15]. MSCs can bind to P-selectin through a fucosylated ligand, which nevertheless is not P-selectin glycoprotein ligand (PSGL)-1 [16]. Thankamony and Sackstein [17] have, however, defined an enzymatic fucosylation procedure which causes the CD44 epitope on MSCs to strongly bind to endothelial E-selectin, resulting in effective rolling of MSCs on endothelial cells and, moreover, extravasation into bone marrow sites. Of the integrins, alpha4beta1 (VLA-4) and alpha5beta1 (VLA-5) have been found to be expressed by MSCs, whereas the beta2 integrins alphaLbeta2 (LFA-1) and alphaMbeta2 (Mac1) could not be detected [15, 16, 18–20] (reviewed in [12, 21]). Interestingly, several chemokine receptors have been found to be expressed on MSCs, including CXCR4, which has been described as a major mediator of the homing and mobilization of hematopoietic cell types [12, 19, 20]. In summary, these findings indicate that MSCs have a deficit with regard to the expressing and/or employing adhesion receptors for coordinated extravasation and tissue-specific homing, as do leukocyte populations.
Emergence of common themes in exploring the biodistribution of MSCs
Subsequent to the first reports on the homing and migration of transplanted MSCs to tissues, additional questions about MSC biodistribution have been addressed, including quantification of MSCs, their preferential homing to several target sites, and the involvement of cues, such as regeneration or inflammation, and the size of MSCs in determining their biodistribution (Table 1).
Table 1
Common themes in MSC biodistribution research
| Theme | Targeted tissues (possible mechanism) | References |
| Increased homing after intra-arterial delivery compared with intravenous delivery? | Kidney | [33, 34] |
| Joints | [32] | |
| Stroke | [30] | |
| Other (many) tissues | [31] | |
| Side effects of intra-arterial versus intravenous delivery? | Incorporation into vessel wall | [23, 35] |
| Obstruction of microvessels | [38] | |
| Vascular occlusion | [39] | |
| Targeting of vessel wall and vessel-associated tissues? | Lungs, lymph nodes, intestine | [47] |
| Targeting of tissues for regeneration | Myocardium | [18, 48–55] |
| Beta1 integrins | [48, 49] | |
| CCL2, monocytes | [52] | |
| Kidney | [33, 56–63] | |
| Gut and liver | [64–67] | |
| Skin | [44, 68–71] | |
| CCL21 | [44] | |
| JAM-A | [68] | |
| Brain | [72–75] | |
| P/E selectin (CD44) | [73] | |
| CXCR4/flk-1/EPO-R | [74] | |
| Homing to bone marrow | Bone marrow | [76–81] |
| HCELL/E-selectin | [15] | |
| Subendothelial localization | [79] | |
| Biodistribution to the immune system? | Macrophages | [37, 41, 42] |
| Dendritic cells | [38] | |
| T cells | [39] | |
| Unknown target cells | ||
| Idoleamine desoxygenase | [43] | |
| Prostaglandin E2 | [37, 41] | |
| Elimination mechanisms? | Antibody formation | [6] |
| Phagocytes | [102] | |
| Influence of radiation on homing? | Increased in brain, heart, bone marrow, and muscles | [43, 82] |
| Homing in malignancies? | Tumor | [83–85, 87–92] |
| Mediated by CCL25 | [88] | |
| Mediated by sodium iodide symporter under the control of RANTES/CCL-5 promoter | [87] | |
| Homed MSCs form tumor-associated fibroblasts | [90] | |
| Formation of microvesicles | Microvesicles may contribute to/be part of MSC biodistribution | [14, 63, 93–97] |
| Mediated by horizontal transfer of microRNAs | [96] |
In many of the earlier studies, the target sites as well as the molecular mechanisms governing the interactions of MSCs with the local environment after transplantation (e.g., endothelial cells, target tissue), such as adhesion molecules or signaling mechanisms, were either not addressed or were analyzed only to a minor degree. Moreover, MSCs were often evaluated by microscopy, a method relatively prone to artifacts. Many studies also did not quantify the numbers of MSCs in target or other tissues. Likewise, only few studies reported on the size of the identified MSCs. Despite this lack of information, other themes have emerged, especially research on cues that may regulate the biodistribution of systemically applied MSCs; these include first pass tissues, specifically the lungs, inflammation, irradiation, sites of hypoxia or repair, and cancer (Table 1). As a result, concepts have been raised which imply an ability of MSCs to migrate to specific sites—e.g., MSCs as an “injury drugstore” for several acute clinical situations [21, 22].
First-line accumulation of intravenously administered MSCs in the lungs
The first hurdle for intravenously transplanted MSCs is the lung capillary bed. After culture expansion, MSCs are relatively large cells with an estimated average size of around 30 µm in suspension (ranging from 16–53 µm) [23]. Their size may also vary depending on the osmolarity of the culture media, passage number, and/or cell density during seeding as well as general culture conditions (two-dimensional versus three-dimensional culture). In comparison with MSCs, hematopoietic stem cells have a much smaller diameter, ranging from 4–12 µm depending on the subfraction analyzed [24, 25]. Therefore, obstructive events during lung passage are expected after intravenous administration of MSCs. Lee et al. [26] presented a kinetic study of MSCs accumulating in murine lungs in which up to 80 % of injected cells were found in the lungs within a few minutes after injection. Moreover, formation of emboli in lung vessels was noted. The MSC signal (an Alu sequence DNA marker) fell exponentially, with a half-life of about 24 h and practically complete disappearance after 4 days [26]. Barbash and colleagues [10] confirmed the detection of the overall MSC load in the lungs using 99mTc-labeled MSCs in a rat model with induced myocardial infarction. Murine MSCs also showed deleterious effects in mice, including post-injection lethality, which was not the case after administration of hMSCs [27]. Interaction of human or murine MSCs with lung endothelial cells was dependent on the suspension medium in which the transplanted cells were administered [27]. Adhesion of the MSCs to endothelial cells was found to involve the integrin ligand vascular cell adhesion molecule (VCAM)-1. When comparing MSCs with mononuclear cells from bone marrow, neural stem cells and multipotent adult progenitor cells, Fischer et al. [28] found that MSCs showed the highest interaction with lung endothelia, which could be inhibited by pretreatment with anti-CD49d antibody. In a study by Kerkelä et al. [29], adhesion of MSCs to lung tissue (probably endothelial cells) was dependent on the enzyme treatment used during harvesting of confluent MSCs in culture before transplantation; after treatment with pronase, MSCs more readily cleared the lungs and could be found in other tissues compared with trypsinization treatment. Taken together, these data indicate an active role of the adhesion molecules VLA-4/VCAM-1 on MSCs/endothelial cells during interaction of MSCs with lung tissue. It remains to be clarified, however, whether this is a passive or active process. Also, relatively little is known about possible adhesion molecules other than VLA-4/VCAM-1 which may be operative in the interaction of MSCs with endothelial cell surfaces in the lung. This includes the fucosylation of CD44 to HCELL, a highly active E-selectin ligand on MSCs, which is relevant in bone marrow endothelia but seemingly did not affect lung interactions [15].
In summary, presently there is strong evidence that accumulation of MSCs in the lungs is a key determining factor for their biodistribution. The major adhesion molecule involved seems to be VLA-4/VCAM1. Still, it is not clear to what degree the findings in animal studies are quantitatively transferable to humans (Table 1).
Biodistribution of MSCs after intra-arterial versus intravenous administration
Studies comparing intra-arterial and intravenous application of MSCs have demonstrated a major association between intravenous application and retention of MSCs in the lungs, and their increased accumulation in therapeutic target tissues after intra-arterial injection. Walczak et al. [30] in a rat transient ischemia stroke model applied two independent detection methods (magnetic resonance imaging and Doppler flowmetry). They demonstrated that higher cerebral engraftment rates are associated with impeded cerebral blood flow, and that intra-arterial delivery may be advantageous in ischemic stroke to deliver MSCs to the site of injury. Mäkelä et al. [31] compared intra-arterial and intravenous administration of MSCs labeled with 99mTc, and also found that the intra-arterial transplantation route has a positive impact on the biodistribution of bone marrow-derived MSCs (BM-MSCs) to peripheral tissues. They found that intra-arterial transplantation decreased the deposition of BM-MSCs in the lungs and increased uptake in other organs, especially in the liver. In a study looking at human adipose tissue-derived MSCs in SCID mice, Toupet et al. [32] showed that 15 % of intra-arterially injected MSCs accumulate in inflamed joints during the first month, and 1.5 % over a longer term of >6 months, also favoring intra-arterial over intravenous application for, in their case, anti-inflammatory MSCs. Therapeutic effects of MSCs in kidney have been generally achieved after intra-arterial delivery [33, 34]. Although more studies will be needed, these data suggest that the intra-arterial route of administration is effective in avoiding pulmonary entrapment of BM-MSCs, and may thus improve the biodistribution and bioavailability of transplanted MSCs in clinically relevant tissues for, e.g., tissue repair.
Interactions of MSCs with the blood vessel wall: integration into the vessel wall or transmigration?
As described above, the majority of intravenously injected MSCs are generally detected in the lungs, and in no other tissue at comparable numbers even at later time points. Some groups asked whether MSCs may directly target vessels or perivascular tissue and investigated the fate of MSCs in and around blood vessels. These studies followed the cells using intravital microscopy and histologic examination in different tissues after intra-arterial [23, 30, 35] administration. In the cremaster muscle intravital microscopy model, Furlani et al. [23] observed that the microcirculation was disturbed, with some MSCs obstructing small vessels. In addition, pulmonary emboli were found. Toma et al. [35] also observed occlusion of microvessels and entrapment of the injected MSCs. Moreover, they observed stable integration of some transplanted cells into the vessel wall. Cui et al. [36] reported a risk of vascular occlusion in their rat stroke infarction model after intra-arterial injection, pointing to the fact that local intravasal entrapment of MSCs may frequently occur, and MSCs may obstruct the microcirculation. Currently, however, we lack conclusive data that MSCs that are entrapped in capillaries and/or are incorporated into the vessel wall or adjacent to endothelial cells would relocate (i.e., “home”) to their main tissue of origin, pericytes.
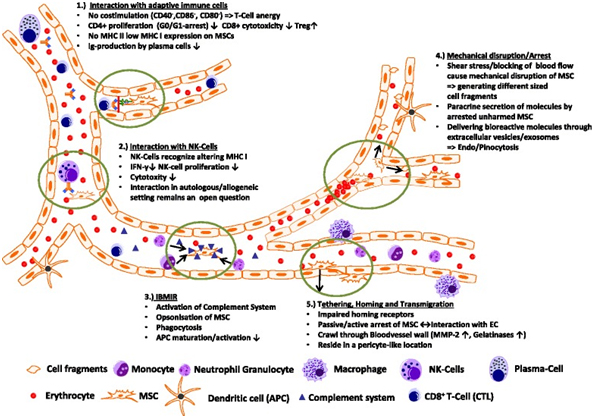
Transplanted MSCs interact with cells of the immune system
Transplanted MSCs have been shown to rapidly interact with immune cell types, which are—at least in part—present also in the bloodstream. In a lung sepsis model, Nemeth et al. [37] observed that MSCs co-localize with lung-resident macrophage cells and induce them to produce anti-inflammatory interleukin (IL)-10 via release of prostaglandin E by MSCs as part of their therapeutic effect. Chiesa et al. [38] showed that interstitial dendritic cells (DCs) decrease their physiological migration from skin to lymph nodes rapidly after intravenous administration of MSCs. They describe that MSCs inhibit Toll-like receptor (TLR)-4-induced activation of DCs, which results in the inhibition of cytokine secretion by DCs, downregulation of adhesion molecules involved in the migration of DCs to the lymph nodes, suppression of DC antigen presentation to CD4+ T cells, and cross-presentation to CD8+ T cells. Akiyama et al. [39] demonstrated that both human and murine MSCs can induce immune suppression by attracting and killing autoreactive T cells through FasL, thereby stimulating transforming growth factor beta production by macrophages and generation of regulatory T cells. The interaction has been shown to involve the secretion of MCP-1 by MSCs. The dying T cells in turn activate macrophages to produce transforming growth factor beta, thus stimulating regulatory T cells and promoting immune tolerance. Possibly, the secretion of anti-inflammatory protein TSG-6 by activated MSCs, which has been described in a zymosan-induced mouse peritonitis model, involves an interaction via TLR2/reduction of NF-?B signaling in resident macrophages [40].
Another type of potential interaction between MSCs and immune cells is suggested by data from Kim et al. [41], who used an in vitro system showing that murine MSCs inhibit functionality of DCs through TLR-4-mediated signals in co-culture with monocytes. During this study, hMSCs revealed a unique immunophenotype of alternatively activated human monocytes which are CD206-high, IL-10-high, IL-6-high, IL12-low, and tumor necrosis factor (TNF)-alpha-low [42]. The immune suppressive effects of MSCs have been shown to depend on induction of indoleamine 2,3-dioxygenase [43], or production of prostaglandin E2 as a main effector to dampen inflammation [37, 44]. These data indirectly support the hypothesis that MSCs interact directly with monocytic and/or antigen-presenting cells in vivo.
The successful therapeutic use of MSCs in patients with severe immune dysregulations, such as graft-versus-host disease after allogeneic hematopoietic stem cell transplantation, has attracted high interest by hematologists (reviewed in [45]). The studies were based on a number of in vitro findings that MSCs can either interact with or affect the function of various types of immune effector cells such as antigen-presenting cells, B or T lymphocytes, or natural killer (NK) cells (reviewed in [46]). In all these studies, identification of MSCs at target sites has been cumbersome, and often no transplanted MSCs were identified. von Bahr et al. [47] reported that MSC donor DNA was detectable at low levels in 8 out of 18 patients in vessel-associated tissues in the patients, including lungs, lymph nodes, and intestine. Detection of MSC donor DNA was negatively correlated with time from infusion to sample collection.
Together, these studies strongly indicate the existence of interactions between transplanted MSCs and cells of the immune system. This way, MSCs also biodistribute to the immune system through contact with different types of leukocytes in the circulation or various tissues such as skin, spleen, and lymph nodes.
Potential mechanisms of elimination of MSCs from the circulation
A relevant aspect of the interaction between transplanted MSCs and immune system cells, in both animal models and humans, is the induction of xenogeneic and allogeneic immune responses, resulting in antibody formation or T-cell responses against the transplanted MSCs. Induction of antibody formation explains the failure to identify transplanted MSCs in patients upon repeated administration of allogeneic MSCs that had been cultured in fetal bovine serum-containing media [6]. Anti-fetal calf serum antibody formation has been demonstrated in patients that did not respond to repeated MSC applications [6]. Elimination of xenogeneic MSCs in some of the animal models studied may occur in ways analogous to those in the allogeneic situation.
Despite the fact that several target tissues of MSCs have been established, there are few data as to the place to which systemically applied MSCs will finally migrate, or where they end up before or when they are eliminated. The fact that the transplanted MSCs are often not detectable at all, or only a small fraction of them is traced, underscores the potential relevance of the lung as a “first pass” tissue, and may indicate an involvement of lung trapping in elimination of MSCs. On the other hand, the fact that MSCs are barely or not at all detectable in patients after transplantation demonstrates that systemic pathways to eliminate transplanted MSCs may be operating in humans, leading to barely detectable long-term engraftment.
Tissue repair situations which provide cues to attract transplanted MSCs
The interactions of MSCs with different types of immune cells point to their ability to respond to signals from the immune system. Since aspects of tissue repair have been associated with (adaptive) immune responses, it is likely that inflammatory and tissue repair signals influence MSC responses in vivo, including their biodistribution.
Myocardial infarction
The VLA-4/VCAM receptor axis has been shown to be involved in MSC migration in myocardial infarction. Pre-treatment of MSCs with TNF-1alpha stimulated migration of MSCs through heart endothelia mediated through VCAM-1, indicating that beta1 integrins are actively involved in this process [48]. Confirming this hypothesis, Ip et al. [49] demonstrated in a murine model that alpha4 integrin is required for migration of MSCs to myocardium, whereas the chemokine receptor CXCR4 was dispensable for the entry of transplanted cells into ischemic tissue.
Intravenously administered MSCs have been observed to, at least transiently, accumulate in areas of myocardial ischemia [18, 50, 51]. To this end, Belema-Bedada et al. [52] employed a transgenic mouse model expressing the monokine CC-chemokine ligand (CCL)2 under a cardiac specific promotor, increasing CCL2 expression in heart muscle. These authors observed that i.v. MSCs accumulate rapidly and selectively in the heart. They showed that the migration of the MSCs to heart is preceded by monocyte emigration to the myocardium, and involves G-protein-coupled receptors, pointing also towards the involvement of chemokine signals. Kraitchman et al. [11] confirmed the accumulation of i.v. MSCs into myocardial infarction areas using a radioimaging tracer and single-photon emission computed tomography in a dog model. Wang et al. [53] traced MSCs at later stages after infarction, and saw markers of newly regenerated cardiomyocytes. It is also not clear whether MSCs steadily incorporate into cardiac tissue. Other studies have failed to detect any homed MSCs in cardiac tissue over the long term (e.g., [54]). Jasmin et al. [55] injected MSCs i.v. after nanoparticle labeling in a model of heart inflammation caused by the Chagas disease parasite Trypanosoma cruzi. They observed that although most MSCs migrated to the lungs, liver and spleen, a few cells homed to the inflamed heart. In conclusion, some mechanisms seem to recruit, mostly transiently, some MSCs to inflamed or ischemic heart, including VLA-4/VCAM-1 and the CCL2 and possibly other chemokine receptor signals.
Kidney damage
Despite the wide range of beneficial effects seen with the therapeutic use of MSCs in animal models, only a few clinical trials have tested the efficacy of MSCs for renal diseases. Reinders and colleagues [56] used intravenous injection of 1?×?106 autologous BM-MSCs/kg in six kidney allograft recipients to dampen rejection of the graft and/or decrease interstitial fibrosis and tubular atrophy. Likewise, Tan et al. investigated autologous BM-MSCs (1–2?×?106/kg) at kidney reperfusion and 2 weeks after application; the incidence of acute rejection decreased and renal function at 1 year improved compared with anti-IL-2 receptor antibody induction therapy [57]. In a clinical phase I safety trial, five patients aged >65 years with underlying renal disease and multiple comorbidities were infused with allogenic MSCs during coronary artery bypass or cardiac surgery. Although the follow-up period was short and one of the patients died, none of the patients required dialysis, supporting the beneficial influence of MSCs on repair of kidney damage [58, 59].
In animal studies, MSCs were also associated with repair of the permeability barrier of the glomerulus in an Alport disease model [60] and improved kidney function in an experimental sepsis mouse model through reprogramming of macrophages via release of prostaglandin E2 [37]. Morigi and colleagues [61, 62] have shown that treatment with murine BM-MSCs (2?×?105 per mouse) in an acute renal failure mouse model induced by cisplatin (a nephrotoxic anti-cancer drug) protected the animals from renal function impairment and tubular injury. Intriguingly, the effects of MSCs in stimulating proliferation and inhibiting apoptosis of tubular epithelial cells in a glycerol-induced acute kidney injury SCID mouse model could also be achieved by using microvesicles derived from hMSCs [63]. In addition to these human studies, several studies demonstrate that MSCs localize within injured kidneys when injected in mice with acute kidney injury (e.g., [34, 63]; reviewed in [58]). The presence of MSCs at later stages of kidney injury or regeneration has not been studied, but the therapeutic benefits have been measured, and intra-arterial injection of MSCs seems to be more favorable [33, 34, 61].
Liver damage
Gholamrezanezhad et al. [14] studied i.v. infused 111In-oxine-labeled MSCs in patients with liver cirrhosis. The radioactivity was first observed to accumulate in the lungs. During the following hours to days, the radioactivity gradually increased in the liver and spleen, with spleen uptake exceeding that in the liver in all patients. In the liver and spleen, radioactivity increased by day 10 post-infusion, whereas residual activity in the lungs decreased approximately tenfold. In contrast, Briquet et al. [64] saw no recruitment of hMSCs to liver damaged by CCl4 intoxication in immune-deficient mice. A study by Zhang et al. [65] indicates that corticosteroids and the SDF-1/CXCR4 axis are involved in MSC migration in a carbon tetrachloride-induced liver fibrosis model. Another liver regeneration model in mice indicated that MSC homing to liver was associated with regeneration, but the mechanisms for this were not investigated [66]. In summary, although many of the published studies have not addressed aspects of MSC biodistribution, there is some evidence for biodistribution to injured or diseased livers, but the underlying mechanisms are mostly unclear.
Gut and skin
Only a few studies have analyzed MSC accumulation in epithelial tissues so far. Inflammatory bowel disease models have addressed homing of i.v. MSCs. Parekaddan et al. [67] demonstrated the presence of MSC-derived signals not only in lungs and spleen but also in the gut of affected animals. Sasaki et al. [44] assessed whether homed MSCs can differentiate into skin cells, including keratinocytes, and whether they could contribute to wound repair. They i.v. injected green fluorescence protein (GFP) transgenic MSCs and identified GFP-positive cells associated with specific markers for keratinocytes, endothelial cells, and pericytes. They attribute the extravasation to inflamed areas to the presence of the chemokine CCL21 in vessels in the inflamed tissue. Still, numbers of detected MSCs in the wounded skin areas were low. MSCs have been found in wound tissues several days after transplantation in animal models [68–71] but their engraftment efficiency ranged from <0.01 % when MSCs were intravenously injected to 3.5 % in a study where MSCs were locally applied. This points to a minor role of i.v. injected MSCs in skin repair. One study reported that, after intravenous injection of GFP transgenic MSCs, keratinocytes, endothelial cells, pericytes and macrophages within the healed wound were all found to be GFP-positive. The authors concluded that they might be derived from donor MSCs [71].
Brain
Some studies have investigated whether transplanted MSCs migrate into inflamed brain tissue. In murine stroke models, MSCs migrated into ischemic areas after intravenous delivery [72, 73]. The latter study mentions that the MSCs are recruited to these sites via endothelial expressed P- and E-selectin, and that CD44 is present on the MSCs. In their rat brain ischemia model, Wei et al. [74] found that i.v. MSCs localize to ischemic zones and deliver neurotrophic factors. This occurs at an increased rate when MSCs have been exposed to hypoxia before transplantation. The extravasation efficiency of the MSCs correlated with increased expression of CXCR4, flk-1 and the erythropoietin receptors, and downregulation of pro-inflammmatory regulators in the homing MSCs. The activity of microglia formation was suppressed in animals after MSC therapy, and NeuN-positive and Glut1-positive cells were increased. Constantin et al. [75] used intravital microscopy in a murine experimental autoimmune encephalitis model. They found, using bioluminescence, accumulation of a subset of transplanted MSCs in inflamed brain venules in inflammatory foci of experimental autoimmune encephalomyelitis 16 and 30 days after transplantation, and showed a role for alpha4 integrin in the migration process of MSCs into brain tissue. Although absolute numbers of transplanted MSCs were not determined and may be low, the results indicate that active inflammation may switch the homing behavior of transplanted MSCs from unspecific entrapment to specific recruitment.
Together, these data indicate that MSCs can migrate into ischemic and proinflammatory regions in certain disease models. Mostly short- (within the first 3 days) and mid-term (3 days to 3 months) homing has been reported, whereas long-term persistence (>3 months) of MSCs is rarely detected. Due to the technologies used to detect transplanted cells, there is only limited evidence to indicate whether the MSCs home as intact cells into their target environments. The data are in favor of transient homing and locally acting MSCs in the investigated pathologies.
Homing of transplanted MSCs to bone marrow
Several decades of clinical and experimental work in the field of bone marrow transplantation have shown that donor type MSCs will generally not engraft in allogeneic hosts, including the precursor cell type for MSCs, fibroblast colony-forming units [76–78]. Rombouts and Ploemacher [79] demonstrated that prolonged time in culture induces a defect in MSCs that affects their engraftment into bone marrow in a classic bone marrow transplantation situation. In contrast, as reported above, Horwitz and colleagues [5, 6] demonstrated that MSCs engraft into bone marrow of children with osteogenesis imperfecta. Possibly, engraftment of MSCs therefore requires a “niche” which is not free in normal bone marrow transplant recipients, but is created in a deficiency state such as the collagen synthase defect found in osteogenesis imperfecta. Follenzi et al. [80] recently demonstrated that mice suffering from hemophilia A, when transplanted with normal healthy total bone marrow cells, show engraftment not only of hematopoietic cells but also of subendothelial MSC-like cells. Interestingly, these MSCs had not been cultured before transplantation. Functional MSCs may, therefore, engraft, at least in the case of certain deficiencies in the transplanted hosts. Interestingly, the group of Horwitz more recently showed that non-plastic-adherent bone marrow cells engraft in a murine model and give rise to osteoprogenitors, which are more potent osteoprogenitors than “classic” plastic-adherent MSCs in mice [81]. This underscores the possibility that the culture period induces the engraftment defect, and that, in addition, cells other than “classic” MSCs can mediate stromal engraftment. On the other hand, “classic” plastic-adherent MSCs have been shown to remain as a source of hematopoietic environment when transplanted into tissues other than bone marrow [1]. In contrast to these findings, the model by Sackstein et al. [15], where an active E-selectin ligand was engineered on the surface of plastic-adherent MSCs, resulted in efficient homing to bone marrow, indicating the possibility of BM-MSCs (or MSCs from other tissue sources) distributing to bone marrow.
Influence of irradiation on migration and biodistribution of MSCs
In a murine study, Francois et al. [43] showed that both total body irradiation and local irradiation (e.g., selective irradiation of abdomen or legs) affected the distribution of i.v. infused hMSCs in NOD/SCID mice compared with untreated animals. Intravenously infused hMSCs were found only in minimal amounts exclusively in the lung, bone marrow, and muscles in non-irradiated control animals. Mice after total body irradiation had increased absolute numbers of hMSCs in brain, heart, bone marrow, and muscles. Moreover, selective radiation of limbs or the abdomen yielded increased engraftment of hMSCs in the exposed skin or muscles than with total body irradiation alone. hMSC engraftment outside the locally irradiated regions was also increased, pointing to both local and systemic effects of irradiation on MSC engraftment. The study did not investigate long-term engraftment. Sémont et al. [82] looked at the engraftment and efficacy of transplanted MSCs in an immunodeficient mouse model of radiation-induced gastrointestinal tract failure. They demonstrated accelerated recovery in the group receiving hMSCs, with decreased apoptosis of epithelial cells and increased proliferation within the small intestinal mucosa. Yet, transplanted MSCs were not detected at significant amounts.
A special case: migration and engraftment of MSCs into tumors
Tumor-associated fibroblasts have been described as a form of MSCs, which are recruited from the MSC pool and are an integral part of the microenvironment of many different solid tumors [83, 84]. Tumor tissue therefore also represents a target for the homing of i.v. injected MSCs. In experimental studies, both beneficial and adverse effects have been reported. Beckermann et al. [85] verified the migration of i.v. MSCs into areas close to the vessel wall in human pancreatic tumors in immunodeficient mice. Alieva et al. [86] followed locally implanted adipose tissue-derived MSCs with a genetic modification induced by lentiviral transduction and traced them by bioluminescence in a glioblastoma model. After incorporation of the transplanted MSCs, administration of gancyclovir activates the thymidine kinase transgene, resulting in death and elimination of the transplanted MSCs and tumor regression. A PECAM-Promotor-driven second transgene as reporter construct served to indicate that the transplanted MSCs can acquire endothelial-like characteristics. Similarly, Knoop et al. [87] used i.v. MSCs expressing sodium iodide symporter under the control of the RANTES/CCL-5 promoter; when loaded with 131I compound these conferred significant anti-tumor effects.
Xu et al. [88], in a myeloma model, showed that MSCs are chemoattracted by the chemokine CCL25, thus supporting myeloma growth. In a Ewing sarcoma nude mouse model, i.v. injected MSCs expressing IL-12 were effective in treating the sarcomas [89]. Interestingly, the transplanted MSCs themselves were not identified, while the secreted IL-12 was. Kidd et al. [90] showed that tumor-associated fibroblasts originating from transplanted MSCs in syngeneic ovarian and breast cancers are recruited from the bone marrow, whereas the bulk of the vascular and fibrovascular stromal cells (pericytes, a-smooth muscle actin-positive myofibroblasts, and endothelial cells) were recruited from adipose tissue. These data indicate a process whereby, once bone marrow homing of transplanted MSCs is established, these MSCs may be (genetically) directed along pre-established pathways of endogenous MSCs that circulate from bone marrow to the tumor. Further work by Grisendi et al. [91] demonstrated that the process of MSC incorporation into tumors implies the formation of epithelial–mesenchymal or endothelial–mesenchymal transitions, and requires the formation of fibroblasts derived from mesenchymal progenitors.
MSCs were also found to enhance angiogenesis, as shown in models of B16 melanoma cells and Lewis lung carcinoma [92]. Co-injection of tumor cells and MSCs led to increased tumor size compared with injection of tumor cells alone. Tumor vessel areas were greater in tumors after co-injection of tumor cells with MSCs than in tumors induced by injection of cancer cells alone. Co-injected MSCs localized close to vascular walls, and also expressed the endothelial marker CD31/PECAM-1.
In conclusion, MSCs show a clear tumor tropism. Many data indicate that they are incorporated into the tumor microenvironment and can stimulate tumor growth. Their biodistribution and tumor tropism, however, may also be exploited to target tumors, e.g., using a suicide transgene approach.
Recent developments: exosomes, microparticles and MSCs
As with many other cell types, MSCs are capable of forming exosomes [63, 93, 94]. Exosomes are small membrane vesicles (40–100 nm in diameter) of endosomal origin derived from MSCs. Exosomes have been found to accumulate in target cells of MSC therapy, such as tubular cells in acute kidney injury [63], or after recovery from traumatic brain injury [95]. In other studies, microvesicles have been found to contain signaling molecules which are hypothesized to be important for MSC-mediated therapeutic effects by horizontal transfer, such as miR-133b in a rodent stroke model [96], or insulin-like growth factor receptor in renal tubular injury [97]. Kordelas et al. [98] administered exosomes isolated from MSCs to a patient with severe graft-versus-host disease; this patient showed marked improvement after the exosome infusion. This field is currently expanding rapidly, and can only be covered briefly by this review. One of the relevant open questions for the biodistribution of MSCs is whether exosomes are indeed formed by intravasally administered MSCs.
Summary: possible ways for MSCs to interact within the local environment of the bloodstream to direct their biodistribution
A summary of the possible ways MSC might interact within the blood circulation is shown in Fig. 1. MSC surface marker profiling has revealed no expression of the co-stimulatory molecules CD40, CD86, and CD80 needed for correct T-cell responses leading to T-cell anergy. In vitro studies also showed that CD4+ T cells in contact with MSCs were arrested in the G1/G0 phase and stopped proliferating whereas regulatory T cell proliferation was favored and IgG production by plasma cells seemed to be affected [46]. In addition, MSCs only express a low amount of major histocompatibility complex (MHC) I and almost no MHC II (except after interferon-? treatment), making them more evasive to NK cell cytoxicity in an allogenic/xenogenic setting. Interactions between NK cells and MSCs in general have been controversial, as discussed by different groups (e.g., [99–101]). MSCs seem to lower NK cell cytoxicity through downregulation of interferon-? expression and production of anti-inflammatory IL-4 and IL 10, but NK cells were associated with the ability to lyse MSCs from allogenic donors [99]. Additionally, the so-called instant blood-mediated inflammatory response might be triggered by the innate immune response caused by tropism of dying MSCs within the blood circulation, resulting in complement activation and opsonization of injected MSCs following uptake of marked MSC cell fragments by primary/secondary phagocytes, as was shown by Moll et al. [102]. Intravital microscopy of MSCs in a cremaster muscle mouse model (our unpublished data) revealed that MSCs are likely to be disrupted by the shear force of the blood flow, resulting in fragmentation of the cell and creation of small extracellular vesicles able to influence paracrine secretion of immunomodulatory molecules or cause phagocytosis of these fragments by macrophages and endothelial cells, subsequently followed by clearance of disrupted MSCs in the liver and spleen within a few days. MSCs that find a niche and survived the journey through the bloodstream might interact actively or passively with the endothelial wall and may extravasate after interacting with the extracellular matrix (e.g., with MMP 2 and gelatinase) and reside in a pericyte-like location in the long term.
Conclusion
The final fate of the bulk of i.v. injected MSCs remains elusive, since preclinical animal studies and some human data have been able to detect only small proportions, if any, of injected MSCs. A number of open questions remain. These include: Which contacts are made between MSCs and other cells upon infusion in the bloodstream and what are the consequences of these? What is the fate of MSCs that do not migrate into inflamed tissue and are there physiological clearance pathways for transplanted MSCs? Given that many therapeutic effects have been observed without detectable MSCs in the target tissues, are intact MSCs therefore relevant for the observed effects?
We believe that further careful analysis of animal disease models, including investigation of the role of mediators such as exosomes, signaling proteins, and microRNAs, will help further advance our understanding of why we have so far not obtained clear answers about how MSCs biodistribute, migrate and home, and how these cells exert their beneficial effects, and what might be the potential of these new insights for the development of further improvements of MSC-derived therapies.
Note
This article is part of a thematic series ‘Mesenchymal Stem/Stromal Cells—An update’. Other articles in this series can be found at http://www.biomedcentral.com/series/mesenchymal
Acknowledgments
The authors thank the team of the Institute of Transfusion Medicine and Immune Hematology, German Red Cross Blood Donor Service, University of Frankfurt for their constant support.
Abbreviations
| BM-MSC | Bone marrow-derived mesenchymal stem/stromal cell |
| DC | Dendritic cell |
| GFP | Green fluorescence protein |
| hMSC | Human mesenchymal stem/stromal cell |
| i.v. | Intravenous/intravenously |
| IL | Interleukin |
| MHC | Major histocompatibility complex |
| MSC | Mesenchymal stem/stromal cell |
| NK | Natural killer |
| TLR | Toll-like receptor |
| TNF | Tumor necrosis factor |
| VCAM | Vascular cell adhesion molecule |
Footnotes
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
All authors read and approved the final manuscript.
References
- Friedenstein AJ, Chailakhyan RK, Latsinik NV, Panasyuk AF, Keiliss-Borok IV. Stromal cells responsible for transferring the micro-environment of the hemopoietic tissues. Cloning in vitro and retransplantation in vivo. Transplantation. 1974;17:331–40. doi: 10.1097/00007890-197404000-00001. [PubMed] [CrossRef] [Google Scholar]
- Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284:143–7. doi: 10.1126/science.284.5411.143. [PubMed] [CrossRef] [Google Scholar]
- Caplan AI, Dennis JE. Mesenchymal stem cells as trophic mediators. J Cell Biochem. 2006;98:1076–84. doi: 10.1002/jcb.20886. [PubMed] [CrossRef] [Google Scholar]
- Caplan AI. Why are MSCs therapeutic? New data: new insight. J Pathol. 2009;217:318–24. doi: 10.1002/path.2469. [PubMed] [CrossRef] [Google Scholar]
- Horwitz EM, Gordon PL, Koo WK, Marx JC, Neel MD, McNall RY, et al. Isolated allogeneic bone marrow-derived mesenchymal cells engraft and stimulate growth in children with osteogenesis imperfecta: implications for cell therapy of bone. Proc Natl Acad Sci U S A. 2002;99:8932–7. doi: 10.1073/pnas.132252399. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Horwitz EM, Prockop DJ, Fitzpatrick LA, Koo WW, Gordon PL, Neel M, et al. Transplantability and therapeutic effects of bone marrow-derived mesenchymal cells in children with osteogenesis imperfecta. Nat Med. 1999;5:309–13. doi: 10.1038/6529. [PubMed] [CrossRef] [Google Scholar]
- Devine SM, Bartholomew A, Mahmud N, Nelson M, Patil S, Hardy W, et al. Mesenchymal stem cells are capable of homing to the bone marrow of non-human primates following systemic infusion. Exp Hematol. 2001;29:244–55. doi: 10.1016/S0301-472X(00)00635-4. [PubMed] [CrossRef] [Google Scholar]
- Devine SM, Cobbs C, Jennings M, Bartholomew A, Hoffman R. Mesenchymal stem cells distribute to a wide range of tissues following systemic infusion into nonhuman primates. Blood. 2003;101:2999–3001. doi: 10.1182/blood-2002-06-1830. [PubMed] [CrossRef] [Google Scholar]
- Gao J, Dennis JE, Muzic RF, Lundberg M, Caplan AI. The dynamic in vivo distribution of bone marrow-derived mesenchymal stem cells after infusion. Cells Tissues Organs. 2001;169:12–20. doi: 10.1159/000047856. [PubMed] [CrossRef] [Google Scholar]
- Barbash IM, Chouraqui P, Baron J, Feinberg MS, Etzion S, Tessone A, et al. Systemic delivery of bone marrow-derived mesenchymal stem cells to the infarcted myocardium: feasibility, cell migration, and body distribution. Circulation. 2003;108:863–8. doi: 10.1161/01.CIR.0000084828.50310.6A. [PubMed] [CrossRef] [Google Scholar]
- Kraitchman D, Tatsumi M, Gilson WD, Ishimori T, Kedziorek D, Walczak P, et al. Dynamic imaging of allogeneic mesenchymal stem cells trafficking to myocardial infarction. Circulation. 2005;112:1451–61. doi: 10.1161/CIRCULATIONAHA.105.537480. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Karp JM, Leng Teo GS. Mesenchymal stem cell homing: the devil is in the details. Cell Stem Cell. 2009;4:206–16. doi: 10.1016/j.stem.2009.02.001. [PubMed] [CrossRef] [Google Scholar]
- Koç ON, Gerson SL, Cooper BW, Dyhouse SM, Haynesworth SE, Caplan AI, et al. Rapid hematopoietic recovery after coinfusion of autologous-blood stem cells and culture-expanded marrow mesenchymal stem cells in advanced breast cancer patients receiving high-dose chemotherapy. J Clin Oncol. 2000;18:307–16. [PubMed] [Google Scholar]
- Gholamrezanezhad A, Mirpour S, Bagheri M, Mohamadnejad M, Alimoghaddam K, Abdolahzadeh L, et al. In vivo tracking of 111In-oxine labeled mesenchymal stem cells following infusion in patients with advanced cirrhosis. Nucl Med Biol. 2011;38:961–7. doi: 10.1016/j.nucmedbio.2011.03.008. [PubMed] [CrossRef] [Google Scholar]
- Sackstein R, Merzaban JS, Cain DW, Dagia NM, Spencer JA, Lin CP, et al. Ex vivo glycan engineering of CD44 programs human multipotent mesenchymal stromal cell trafficking to bone. Nat Med. 2008;14:181–7. doi: 10.1038/nm1703. [PubMed] [CrossRef] [Google Scholar]
- Ruster B, Gottig S, Ludwig RJ, Bistrian R, Muller S, Seifried E, et al. Mesenchymal stem cells display coordinated rolling and adhesion behavior on endothelial cells. Blood. 2006;108:3938–44. doi: 10.1182/blood-2006-05-025098. [PubMed] [CrossRef] [Google Scholar]
- Thankamony SP, Sackstein R. Enforced hematopoietic cell E- and L-selectin ligand (HCELL) expression primes transendothelial migration of human mesenchymal stem cells. Proc Natl Acad Sci U S A. 2011;108:2258–63. doi: 10.1073/pnas.1018064108. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Schenk S, Mal N, Finan A, Zhang M, Kiedrowski M, Popovic Z, et al. Monocyte chemotactic protein-3 is a myocardial MSC homing factor. Stem Cells. 2007;25:245–51. doi: 10.1634/stemcells.2006-0293. [PubMed] [CrossRef] [Google Scholar]
- Shi M, Li J, Liao L, Chen B, Li B, Chen L, et al. Regulation of CXCR4 expression in human mesenchymal stem cells by cytokine treatment: role in homing efficiency in NOD/SCID mice. Haematologica. 2007;92:897–904. doi: 10.3324/haematol.10669. [PubMed] [CrossRef] [Google Scholar]
- Potapova IA, Brink PR, Cohen IS, Doronin SV. Culturing of human mesenchymal stem cells as three-dimensional aggregates induces functional expression of CXCR4 that regulates adhesion to endothelial cells. J Biol Chem. 2008;283:13100–7. doi: 10.1074/jbc.M800184200. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Caplan A, Correa D. The MSC: an injury drugstore. Cell Stem Cell. 2011;9:11–5. doi: 10.1016/j.stem.2011.06.008. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Stappenbeck TS, Miyoshi H. The role of stromal stem cells in tissue regeneration and wound repair. Science. 2009;324:1666–9. doi: 10.1126/science.1172687. [PubMed] [CrossRef] [Google Scholar]
- Furlani D, Ugurlucan M, Ong L, Bieback K, Pittermann E, Westien I, et al. Is the intravascular administration of mesenchymal stem cells safe? Mesenchymal stem cells and intravital microscopy. Microvasc Res. 2009;77:370–6. doi: 10.1016/j.mvr.2009.02.001. [PubMed] [CrossRef] [Google Scholar]
- Sharma S, Krishan A. Hematopoietic Stem Cells. In: Krishan A, Krishnamurthy H, Totey S, editors. Applications of flow cytometry in stem cell research and tissue regeneration. New Jersey: Wiley-Blackwell; 2010. pp. 103–14. [Google Scholar]
- Radley JM, Ellis S, Palatsides M, Williams B, Bertoncello I. Ultrastructure of primitive hematopoietic stem cells isolated using probes of functional status. Exp Hematol. 1999;27:365–9. doi: 10.1016/S0301-472X(98)00017-4. [PubMed] [CrossRef] [Google Scholar]
- Lee RH, Pulin AA, Seo MJ, Kota DJ, Ylostalo J, Larson BL, et al. Intravenous hMSCs improve myocardial infarction in mice because cells embolized in lung are activated to secrete the anti-inflammatory protein TSG-6. Cell Stem Cell. 2009;5:54–63. doi: 10.1016/j.stem.2009.05.003. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Deak E, Rüster B, Keller L, Eckert K, Fichtner I, Seifried E, et al. Suspension medium influences interaction of mesenchymal stromal cells with endothelium and pulmonary toxicity after transplantation in mice. Cytotherapy. 2010;12:260–4. doi: 10.3109/14653240903401840. [PubMed] [CrossRef] [Google Scholar]
- Fischer UM, Harting MT, Jimenez F, Monzon-Posadas WO, Xue H, Savitz SI, et al. Pulmonary passage is a major obstacle for intravenous stem cell delivery: the pulmonary first-pass effect. Stem Cells Dev. 2009;18(5):683–92. doi: 10.1089/scd.2008.0253. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Kerkelä E, Hakkarainen T, Mäkelä T, Raki M, Kambur O, Kilpinen L, et al. Transient proteolytic modification of mesenchymal stromal cells increases lung clearance rate and targeting to injured tissue. Stem Cells Transl Med. 2013;2(7):510–20. doi: 10.5966/sctm.2012-0187. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Walczak P, Zhang J, Gilad AA, Kedziorek DA, Ruiz-Cabello J, Young RG, et al. Dual-modality monitoring of targeted intraarterial delivery of mesenchymal stem cells after transient ischemia. Stroke. 2008;39:1569–74. doi: 10.1161/STROKEAHA.107.502047. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Mäkelä T, Takalo R, Arvola O, Haapanen H, Yannopoulos F, Blanco R, et al. Safety and biodistribution study of bone marrow-derived mesenchymal stromal cells and mononuclear cells and the impact of the administration route in an intact porcine model. Cytotherapy. 2015;17:392–402. doi: 10.1016/j.jcyt.2014.12.004. [PubMed] [CrossRef] [Google Scholar]
- Toupet K, Maumus M, Luz-Crawford P, Lombardo E, Lopez-Belmonte J, van Lent P, et al. Survival and biodistribution of xenogenic adipose mesenchymal stem cells is not affected by the degree of inflammation in arthritis. PLoS One. 2015;10:e0114962. doi: 10.1371/journal.pone.0114962. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Tögel F, Yang Y, Zhang P, Hu Z, Westenfelder C. Bioluminescence imaging to monitor the in vivo distribution of administered mesenchymal stem cells in acute kidney injury. Am J Physiol Renal Physiol. 2008;295:F315–21. doi: 10.1152/ajprenal.00098.2008. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Morigi M, De Coppi P. Cell therapy for kidney injury: different options and mechanisms—mesenchymal and amniotic fluid stem cells. Nephron Exp Nephrol. 2014;126:59. doi: 10.1159/000360667. [PubMed] [CrossRef] [Google Scholar]
- Toma C, Wagner WR, Bowry S, Schwartz A, Villanueva F. Fate of culture-expanded mesenchymal stem cells in the microvasculature: in vivo observations of cell kinetics. Circ Res. 2009;104(3):398–402. doi: 10.1161/CIRCRESAHA.108.187724. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Cui LL, Kerkelä E, Bakreen A, Nitzsche F, Andrzejewska A, Nowakowski A, et al. The cerebral embolism evoked by intraaterial delivery of allogeneic bone marrow mesenchymal stem cells in rats is related to cell dose and infusion velocity. Stem Cell Res Ther. 2015;6:11. doi: 10.1186/scrt544. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Nemeth K, Leelahavanichkul A, Yuen PS, Mayer B, Parmelee A, Doi K, et al. Bone marrow stromal cells attenuate sepsis via prostaglandin E(2)-dependent reprogramming of host macrophages to increase their interleukin-10 production. Nat Med. 2009;15:42–9. doi: 10.1038/nm.1905. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Chiesa S, Morbelli S, Morando S, Massollo M, Marini C, Bertoni A, et al. Mesenchymal stem cells impair in vivo T-cell priming by dendritic cells. Proc Natl Acad Sci U S A. 2011;108:17384–9. doi: 10.1073/pnas.1103650108. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Akiyama K, Chen C, Wang D, Xu X, Qu C, Yamaza T, et al. Mesenchymal stem cell-induced immunoregulation involves Fas ligand/Fas-mediated T cell apoptosis. Cell Stem Cell. 2012;10(5):544–55. doi: 10.1016/j.stem.2012.03.007. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Choi H, Lee RH, Bazhanov N, Oh JY, Prockop DJ. Anti-inflammatory protein TSG-6 secreted by activated MSCs attenuates zymosan-induced mouse peritonitis by decreasing TLR2/NF-?B signaling in resident macrophages. Blood. 2011;118:330–8. doi: 10.1182/blood-2010-12-327353. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Kim J, Hematti P. Mesenchymal stem celleducated macrophages: a novel type of alternatively activated macrophages. Exp Hematol. 2009;37:1445–53. doi: 10.1016/j.exphem.2009.09.004. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Maggini J, Mirkin G, Bognanni I, Holmberg J, Piazzón IM, Nepomnaschy I, et al. Mouse bone marrow-derived mesenchymal stromal cells turn activated macrophages into a regulatory-like profile. PLoS One. 2010;5(2):e9252. doi: 10.1371/journal.pone.0009252. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Francois M, Romieu-Mourez R, Li M, Galipeau J. Human MSC suppression correlates with cytokine induction of indoleamine 2,3-dioxygenase and bystander M2 macrophage differentiation. Mol Ther. 2011;20:187–95. doi: 10.1038/mt.2011.189. [PubMed] [CrossRef] [Google Scholar]
- Sasaki M, Abe R, Fujita Y, Ando S, Inokuma D, Shimizu H. Mesenchymal stem cells are recruited into wounded skin and contribute to wound repair by transdifferentiation into multiple skin cell type. J Immunol. 2008;180:2581–7. doi: 10.4049/jimmunol.180.4.2581. [PubMed] [CrossRef] [Google Scholar]
- Le Blanc K, Mougiakanos D. Multipotent mesenchymal stromal cells and the innate immune system. Nat Rev Immunol. 2012;12:383–96. doi: 10.1038/nri3209. [PubMed] [CrossRef] [Google Scholar]
- Nauta AJ, Fibbe WE. Immunomodulatory properties of mesenchymal stromal cells. Blood. 2007;110:3499–506. doi: 10.1182/blood-2007-02-069716. [PubMed] [CrossRef] [Google Scholar]
- von Bahr L, Batsis I, Moll G, Hägg M, Szakos A, Sundberg B, et al. Analysis of tissues following mesenchymal stromal cell therapy in humans indicates limited long-term engraftment and no ectopic tissue formation. Stem Cells. 2012;30:1575–8. doi: 10.1002/stem.1118. [PubMed] [CrossRef] [Google Scholar]
- Segers VFM, Van Riet I, Andries LJ, Lemmens K, Demolder MJ, De Becker AJML, et al. Mesenchymal stem cell adhesion to cardiac microvascular endothelium: activators and mechanisms. Am J Physiol Heart Circ Physiol. 2006;290:H1370–7. doi: 10.1152/ajpheart.00523.2005. [PubMed] [CrossRef] [Google Scholar]
- Ip JE, Wu Y, Huang J, Zhang L, Pratt RE, Dzau VJ. Mesenchymal stem cells use integrin beta1 not CXC chemokine receptor 4 for myocardial migration and engraftment. Mol Biol Cell. 2007;18:2873–82. doi: 10.1091/mbc.E07-02-0166. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Wu Y, Zhao RC. The role of chemokines in mesenchymal stem cell homing to myocardium. Stem Cell Rev. 2012;8:243–50. doi: 10.1007/s12015-011-9293-z. [PubMed] [CrossRef] [Google Scholar]
- Zhang M, Mal N, Kiedrowski M, Chacko M, Askari AT, Popovic ZB, et al. SDF-1 expression by mesenchymal stem cells results in trophic support of cardiac myocytes after myocardial infarction. FASEB J. 2007;21:3197–207. doi: 10.1096/fj.06-6558com. [PubMed] [CrossRef] [Google Scholar]
- Belema-Bedada F, Uchida S, Martire A, Kostin S, Braun T. Efficient homing of multipotent adult MSCs depends on FROUNT-mediated clustering of CCR2. Cell Stem Cell. 2008;2:566–75. doi: 10.1016/j.stem.2008.03.003. [PubMed] [CrossRef] [Google Scholar]
- Wang Y, Zhang D, Ashraf M, Zhao T, Huang W, Ashraf A, et al. Combining neuropeptide Y and mesenchymal stem cells reverses remodeling after myocardial infarction. Am J Physiol Heart Circ Physiol. 2010;298:H275–86. doi: 10.1152/ajpheart.00765.2009. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Laurila JP, Laatikainen L, Castellone MD, Trivedi P, Heikkila J, Hinkkanen A, et al. Human embryonic stem cell-derived mesenchymal stromal cell transplantation in a rat hind limb injury model. Cytotherapy. 2009;11:726–37. doi: 10.3109/14653240903067299. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Jasmin, Jelicks LA, Tanowitz HB, Peters VM, Mendez-Otero R, de Carvalho AC C, et al. Molecular imaging, biodistribution and efficacy of mesenchymal bone marrow cell therapy in a mouse model of Chagas disease. Microbes Infect. 2014;16:923–35. doi: 10.1016/j.micinf.2014.08.016. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Reinders ME, de Fijter JW, Roelofs H, Bajema IM, de Vries DK, Schaapherder AF, et al. Autologous bone marrow-derived mesenchymal stromal cells for the treatment of allograft rejection after renal transplantation: results of a phase I study. Stem Cells Transl Med. 2013;2:107–11. doi: 10.5966/sctm.2012-0114. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Tan J, Wu W, Xu X, Liao L, Zheng F, Messinger S, et al. Induction therapy with autologous mesenchymal stem cells in living-related kidney transplants: a randomized controlled trial. JAMA. 2012;307:1169–77. doi: 10.1001/jama.2012.316. [PubMed] [CrossRef] [Google Scholar]
- Humphreys BD, Bonventre JV. Mesenchymal stem cells in acute kidney injury. Annu Rev Med. 2008;59:311–25. doi: 10.1146/annurev.med.59.061506.154239. [PubMed] [CrossRef] [Google Scholar]
- Gooch A, Doty J, Flores J, Swenson L, Toegel FE, Reiss GR, et al. Initial report on a phase I clinical trial: prevention and treatment of post-operative acute kidney injury with allogeneic mesenchymal stem cells in patients who require on-pump cardiac surgery. Cell Ther Transplant. 2008;1:e.000028.01. [Google Scholar]
- Sugimoto H, Mundel TM, Sund M. Bone-marrow-derived stem cells repair basement membrane collagen defects and reverse genetic kidney disease. Proc Natl Acad Sci U S A. 2006;103:7321–6. doi: 10.1073/pnas.0601436103. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Morigi M, Imberti B, Zoja C, Corna D, Tomasoni S, Abbate M, et al. Mesenchymal stem cells are renotropic, helping to repair the kidney and improve function in acute renal failure. J Am Soc Nephrol. 2004;15(7):1794–804. doi: 10.1097/01.ASN.0000128974.07460.34. [PubMed] [CrossRef] [Google Scholar]
- Imberti B, Morigi M, Tomasoni S, Rota C, Corna D, Longaretti L, et al. Insulin-like growth factor-1 sustains stem cell mediated renal repair. J Am Soc Nephrol. 2007;18:2921–8. doi: 10.1681/ASN.2006121318. [PubMed] [CrossRef] [Google Scholar]
- Bruno S, Grange C, Deregibus MC, Calogero RA, Saviozzi S, Collino F, et al. Mesenchymal stem cell-derived microvesicles protect against acute tubular injury. J Am Soc Nephrol. 2009;20:1053–67. doi: 10.1681/ASN.2008070798. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Briquet A, Grégoire C, Comblain F, Servais L, Zeddou M, Lechanteur C, et al. Human bone marrow, umbilical cord or liver mesenchymal stromal cells fail to improve liver function in a model of CCl4-induced liver damage in NOD/SCID/IL-2R?(null) mice. Cytotherapy. 2014;16:1511–8. doi: 10.1016/j.jcyt.2014.07.003. [PubMed] [CrossRef] [Google Scholar]
- Zhang S, Lv C, Yang X, Han Z, Zhang S, Zhang J, et al. Corticosterone mediates the inhibitory effect of restraint stress on the migration of mesenchymal stem cell to carbon tetrachloride-induced fibrotic liver by downregulating CXCR4/7 expression. Stem Cells Dev. 2015;24(5):587–96. doi: 10.1089/scd.2014.0243. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Li Q, Zhou X, Shi Y, Li J, Zheng L, Cui L, et al. In vivo tracking and comparison of the therapeutic effects of MSCs and HSCs for liver injury. PLoS One. 2013;8(4) doi: 10.1371/journal.pone.0062363. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Parekkadan B, Upadhyay R, Dunham J, Iwamoto Y, Mizoguchi E, Mizoguchi A, et al. Bone marrow stromal cell transplants prevent experimental enterocolitis and require host CD11b?+?splenocytes. Gastroenterology. 2011;140:966–75. doi: 10.1053/j.gastro.2010.10.013. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Wu M, Ji S, Xiao S, Kong Z, Fang H, Zhang Y, et al. JAM-A promotes wound healing by enhancing both homing and secretion activities of mesenchymal stem cells. Clin Sci (Lond). 2015. [Epub ahead of print]. [PubMed]
- Kidd S, Spaeth E, Dembinski JL, Dietrich M, Watson K, Klopp A, et al. Direct evidence of mesenchymal stem cell tropism for tumor and wounding microenvironments using in vivo bioluminescent imaging. Stem Cells. 2009;27:2614. doi: 10.1002/stem.187. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Fathke C, Wilson L, Hutter J, Kapoor V, Smith A, Hocking A, et al. Contribution of bone marrow-derived cells to skin: collagen deposition and wound repair. Stem Cells. 2004;22:812. doi: 10.1634/stemcells.22-5-812. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Wu Y, Chen L, Scott PG, Tredget EE. Mesenchymal stem cells enhance wound healing through differentiation and angiogenesis. Stem Cells. 2007;25:2648. doi: 10.1634/stemcells.2007-0226. [PubMed] [CrossRef] [Google Scholar]
- Wu J, Sun Z, Sun H-S, Wu J, Weisel RD, Keating A, et al. Intravenously administered bone marrow cells migrate to damaged brain tissue and improve neural function in ischemic rats. Cell Transplant. 2008;16:993–1005. doi: 10.3727/000000007783472435. [PubMed] [CrossRef] [Google Scholar]
- Yilmaz G, Vital S, Yilmaz CE, Stokes KY, Alexander JS, Granger DN. Selectin-mediated recruitment of bone marrow stromal cells in the postischemic cerebral microvasculature. Stroke. 2011;42:806–11. doi: 10.1161/STROKEAHA.110.597088. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Wei L, Fraser JL, Lu ZY, Hu X, Yu SP. Transplantation of hypoxia preconditioned bone marrow mesenchymal stem cells enhances angiogenesis and neurogenesis after cerebral ischemia in rats. Neurobiol Dis. 2012;46:635–45. doi: 10.1016/j.nbd.2012.03.002. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Constantin G, Marconi S, Rossi B, Angiari S, Calderan L, Anghileri E, et al. Adipose-derived mesenchymal stem cells ameliorate chronic experimental autoimmune encephalomyelitis. Stem Cells. 2009;27:2624–35. doi: 10.1002/stem.194. [PubMed] [CrossRef] [Google Scholar]
- Simmons PJ, Przepiorka D, Thomas ED, Torok-Storb B. Host origin of marrow stromal cells following allogeneic bone marrow transplantation. Nature. 1987;328:429–32. doi: 10.1038/328429a0. [PubMed] [CrossRef] [Google Scholar]
- Cilloni D, Carlo-Stella C, Falzetti F, Sammarelli G, Regazzi E, Colla S, et al. Limited engraftment capacity of bone marrow-derived mesenchymal cells following T-cell-depleted hematopoietic stem cell transplantation. Blood. 2000;96:3637–43. [PubMed] [Google Scholar]
- Rieger K, Marinets O, Fietz T, Körper S, Sommer D, Mücke C, et al. Mesenchymal stem cells remain of host origin even a long time after allogeneic peripheral blood stem cell or bone marrow transplantation. Exp Hematol. 2005;33:605–11. doi: 10.1016/j.exphem.2005.02.004. [PubMed] [CrossRef] [Google Scholar]
- Rombouts WJ, Ploemacher RE. Primary murine MSC show highly efficient homing to the bone marrow but lose homing ability following culture. Leukemia. 2003;17:160–70. doi: 10.1038/sj.leu.2402763. [PubMed] [CrossRef] [Google Scholar]
- Follenzi A, Raut S, Merlin S, Sarkar R, Gupta S. Role of bone marrow transplantation for correcting hemophilia A in mice. Blood. 2012;119:5532–42. doi: 10.1182/blood-2011-07-367680. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Otsuru S, Gordon PL, Shimono K, Jethva R, Marino R, Phillips CL, et al. Transplanted bone marrow mononuclear cells and MSCs impart clinical benefit to children with osteogenesis imperfecta through different mechanisms. Blood. 2012;120:1933–41. doi: 10.1182/blood-2011-12-400085. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Sémont A, Mouiseddine M, François A, Demarquay C, Mathieu N, Chapel A, et al. Mesenchymal stem cells improve small intestinal integrity through regulation of endogenous epithelial cell homeostasis. Cell Death Differ. 2010;17:952–61. doi: 10.1038/cdd.2009.187. [PubMed] [CrossRef] [Google Scholar]
- Fakhrejahani E, Toi M. Tumor angiogenesis: pericytes and maturation are not to be ignored. J Oncol. 2012;2012:261750. doi: 10.1155/2012/261750. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- D’souza N, Burns JS, Grisendi G, Candini O, Veronesi E, Piccinno S, et al. MSC and tumors: homing, differentiation, and secretion influence therapeutic potential. Adv Biochem Eng Biotechnol. 2013;130:209–66. [PubMed] [Google Scholar]
- Beckermann BM, Kallifatidis G, Groth A, Frommhold D, Apel A, Mattern J, et al. VEGF expression by mesenchymal stem cells contributes to angiogenesis in pancreatic carcinoma. Br J Cancer. 2008;99:622–31. doi: 10.1038/sj.bjc.6604508. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Alieva M, Bagó JR, Aguilar E, Soler-Botija C, Vila OF, Molet J, et al. Glioblastoma therapy with cytotoxic mesenchymal stromal cells optimized by bioluminescence imaging of tumor and therapeutic cell response. PLoS One. 2012;7:e35148. doi: 10.1371/journal.pone.0035148. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Knoop K, Schwenk N, Schmohl K, Müller A, Zach C, Cyran C, et al. Mesenchymal stem cell (MSC)-mediated, tumor stroma-targeted radioiodine therapy of metastatic colon cancer using the sodium iodide symporter as theranostic gene. J Nucl Med. 2015;pii:jnumed.114.146662. [PubMed]
- Xu S, Menu E, De Becker A, Van Camp B, Vanderkerken K, Van Riet I. Bone marrow-derived mesenchymal stromal cells are attracted by multiple myeloma cell-produced chemokine CCL25 and favor myeloma cell growth in vitro and in vivo. Stem Cells. 2012;30:266–79. doi: 10.1002/stem.787. [PubMed] [CrossRef] [Google Scholar]
- Duan X, Guan H, Cao Y, Kleinerman ES. Murine bone marrow-derived mesenchymal stem cells as vehicles for interleukin-12 gene delivery into Ewing sarcoma tumors. Cancer. 2009;115:13–22. doi: 10.1002/cncr.24013. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Kidd S, Spaeth E, Watson K, Burks J, Lu H, Klopp A, et al. Origins of the tumor microenvironment: quantitative assessment of adipose-derived and bone marrow-derived stroma. PLoS One. 2012;7:e30563. doi: 10.1371/journal.pone.0030563. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Grisendi G, Bussolari R, Veronesi E, Piccinno S, Burns JS, De Santis G, et al. Understanding tumor-stroma interplays for targeted therapies by armed mesenchymal stromal progenitors: the Mesenkillers. Am J Cancer Res. 2011;1:787–805. [PMC free article] [PubMed] [Google Scholar]
- Suzuki K, Sun R, Origuchi M, Kanehira M, Takahata T, Itoh J, et al. Mesenchymal stromal cells promote tumor growth through the enhancement of neovascularization. Mol Med. 2011;17:579–87. doi: 10.2119/molmed.2010.00157. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Yu B, Zhang X, Li X. Exosomes derived from mesenchymal stem cells. Int J Mol Sci. 2014;15:4142–57. doi: 10.3390/ijms15034142. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Akyurekli C, Le Y, Richardson RB, Fergusson D, Tay J, Allan DS. A systematic review of preclinical studies on the therapeutic potential of mesenchymal stromal cell-derived microvesicles. Stem Cell Rev. 2015;11:150–60. doi: 10.1007/s12015-014-9545-9. [PubMed] [CrossRef] [Google Scholar]
- Zhang Y, Chopp M, Meng Y, Katakowski M, Xin H, Mahmood A, et al. Effect of exosomes derived from multipluripotent mesenchymal stromal cells on functional recovery and neurovascular plasticity in rats after traumatic brain injury. J Neurosurg. 2015;16:1–12. [PMC free article] [PubMed] [Google Scholar]
- Xin H, Li Y, Liu Z, Wang X, Shang X, Cui Y, et al. MiR-133b promotes neural plasticity and functional recovery after treatment of stroke with multipotent mesenchymal stromal cells in rats via transfer of exosome-enriched extracellular particles. Stem Cells. 2013;31:2737–46. doi: 10.1002/stem.1409. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Tomasoni S, Longaretti L, Rota C, Morigi M, Conti S, Gotti E, et al. Transfer of growth factor receptor mRNA via exosomes unravels the regenerative effect of mesenchymal stem cells. Stem Cells Dev. 2013;22:772–80. doi: 10.1089/scd.2012.0266. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Kordelas L, Rebmann V, Ludwig AK, Radtke S, Ruesing J, Doeppner TR, et al. MSC-derived exosomes: a novel tool to treat therapy-refractory graft-versus-host disease. Leukemia. 2014;28:970–3. [PubMed] [Google Scholar]
- Spaggiari GM, Capobianco A, Becchetti S, Mingari MC, Moretta L. Mesenchymal stem cell-natural killer cell interactions: evidence that activated NK cells are capable of killing MSCs, whereas MSCs can inhibit IL-2-induced NK-cell proliferation. Blood. 2006;107:1484–90. doi: 10.1182/blood-2005-07-2775. [PubMed] [CrossRef] [Google Scholar]
- Giuliani M, Bennaceur-Griscelli A, Nanbakhsk A, Oudrhiri N, Chouaib S, Azzarone B, et al. TLR ligands stimulation protects MSC from NK killing. Stem Cells. 2014;32:290–300. doi: 10.1002/stem.1563. [PubMed] [CrossRef] [Google Scholar]
- Reinders ME, Hoogduijn MJ. NK cells and MSCs: possible implications for MSC therapy in renal transplantation. J Stem Cell Res Ther. 2014;4:10000166. doi: 10.4172/2157-7633.1000166. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Moll G, Rasmusson-Duprez I, Von Bahr L, Conolly-Andersen AM, Elgue G, Funke L, et al. Are therapeutic human mesenchymal stromal cells compatible with human blood. Stem Cells. 2012;30:1565–74. doi: 10.1002/stem.1111. [PubMed] [CrossRef] [Google Scholar]
Articles from Stem Cell Research & Therapy are provided here courtesy of BioMed Central
- Published in Corporate News / Blog
The FDA Vs Stem Cell Treatments In The US
The FDA Vs Stem Cell Treatments In The US
Stem cell therapy has become a popular type of treatment for a number of conditions, including to assist in the regeneration of injured tissue or diseases cells in the human body. This therapy is classified as regenerative medicine. Several advancements have already been made in this medical field, and many had experienced positive results when they underwent stem cell therapy to assist in the treatment of degenerative conditions.
While stem cell therapy has been proven to provide an effective protocol in the recovery of stroke, neurological issues, and even to assist in repairing the damage that was dealt by a heart attack, recent reports have caused many to view these treatments in a negative perspective. This is due to the warnings that were recently issued by the FDA. Even though some have experienced side-effects due to the use of stem cell therapy, it is important to note that there are facilities within the US that can provide professional services that help to minimize the risk of these adverse events.
The FDA On Stem Cell Therapy In The United States
The Food and Drug Administration of the United States is involved in the analyzing and approval of medications and medical procedures that may be offered to patients in the country. To date, the FDA has shown some interest in stem cell therapy, providing approval for several clinical trials that looked at how effective this treatment is in providing a regenerating effect on the impact that certain conditions has in the human body. Only one particular treatment that involves stem cell therapy has been officially approved by the Food and Drug Administration, however.
Within the last few months, we have seen several concerning publications made by the FDA regarding stem cell therapies, reducing the hope that some patients might have gained.
On the 25th of June, 2019, the FDA published a statement that described a permanent injunction that will occur with the many stem cell clinics in the country. The FDA has stated that the action taken from their side is to assist in providing a layer of protection for patients, due to the risks involved with undergoing treatments that utilize products that have not gone through any type of approval process.
The specific company that was targeted in this statement and by the actions that were taken by the FDA is known as US Stem Cell Clinic LLC. The court ruled in favor of the FDA and US government earlier in June, finding the defendants, being US Stem Cell Clinic LLC, guilty of the claims that were made against them.
An earlier publication also confirmed that the Federal court has decided to rule the misbranding of the stem cell products released by the US Stem Cell Clinics company as a violation of laws that have been implemented to protect the people of the country.
News publications have also announced that the regulations and guidelines that have now been set out by the FDA might cause a significant decline in the availability of stem cell treatments to patients in the United States. The companies that have been targeted through these particular actions that the FDA has taken are primarily those that have been marketing products that have not gone through approval phases – and these products have been found to put the patient’s health at risk.
The Role Of Stem Cell Therapy In Disease
While some companies have been found guilty of using unapproved and even misleading products on patients, promoted as stem cell therapy, it is important to consider the reality of the situation as well. Stem cell therapy often referred to simply as Cell therapy, has been proven as a successful regime in the treatment of several conditions. Many of the conditions that have shown improvement with the use of this therapy were previously considered difficult, or sometimes even impossible, to effectively treat.
By 2012, the Worldwide Network for Blood and Marrow Transplantation, also known as the WBMT, announced that a total of one million stem cell therapy procedures had been done throughout the world. This was a significant milestone, and these procedures have helped to save a countless number of lives. Just in this report, the WBMT refers back to a case of Marta, a girl from Madrid, who had received stem cell therapy at a young age. This treatment was provided to Marta after she was diagnosed with Leukemia. In 2002, Marta was able to overcome Leukemia, thanks to the cell therapy that was provided to her.
While lymphoma and leukemia are the conditions where patients most frequently seek a clinic that can perform stem cell therapy on them, there are many other conditions that are treated with this procedure today. The perfection of the therapy has led to a treatment that can assist in reducing the effects of over 70 diseases that may otherwise have a significant impact on the human body.
GSCG Expands To Cancun, Meets The Demands Of US Patients
The Global Stem Cells Group had recently announced the opening of a new office that they have decided to establish in Cancun, Mexico. The decision was driven by the increasing demand for cell therapy procedures by patients in the United States and by the fact that there is currently a lack of facilities that are able to provide these individuals with the professional and quality services needed. Doctors specializing in regenerative medicine are also having a hard time fulfilling the needs of patients who wish to consider cell therapy as a potential treatment option.
This is not the only local office that GSCG now operates in Cancun, as the company now has two facilities within this particular location. These offices include a stem cells laboratory, as well as a general medical facility.
Over the years, GSCG has become an established name in the stem cell field, providing thousands of doctors the opportunity to offer cell therapy as a treatment option for patients with degenerative diseases. The use of this therapy may assist in the regeneration of damaged and diseased tissues in the patient’s body.
Even though a previous office had established a presence for GSCG in Cancun, it should be noted that the company announced that the opening of the new office means they have now officially created a permanent presence for the brand in the area.
The GSCG’s new facility in Cancun is equipped with the latest advancements that have been made in the field and provide quality services that ensure patients have a trusted medical facility where their patients can undergo cell therapy as part of a regenerative medicine treatment plan.
At the moment, doctors who are in the field of regenerative medicine within the United States can turn to this facility to offer their patients an additional treatment option, apart from the standard pharmaceutical protocols that have been established. The stem cell laboratory can assist in the analyzing of patient data and also in the process of finding a matching donor for the person who is in need of stem cell treatment.
GSCG’s facility is able to assist with the culturing of expanded autonomous cells, as well as other types of cells that may be utilized in the process of stem cell therapy. A detailed approach is taken, considering the age of the donor, as this would also provide an overview of the autonomous stem cell age. The patient being treated is also taken into close consideration, as there are different methods of introducing these stem cells to the body for enhanced efficacy.
A more aggressive method is often needed in those patients with conditions such as autism, Alzheimer’s disease, and kidney-related conditions. Other conditions can also be treated, such as pulmonary diseases, Parkinson’s disease. In cases of a stroke, the vascular arteries, along with the carotid artery, is targeted with the therapy for a more effective approach to assisting with the regeneration of damaged structures in the patient’s brain. This ultimately leads to a significant improvement in the delivery of stem cells to the patient’s body, which could enhance the overall efficacy expressed by the treatment.
When these procedures are provided to the patient through the GSCG, an appropriate ICU unit is made available to ensure the patient is provided with adequate care during the operation and recovery of the procedure.
The utilization of recent technological advancements at laboratories facilitated by GSCG can also assist in providing a more economical approach to cell therapy. The utilization of allogenic stem cell procedures can be addressed using techniques that will essentially offer a reduction in the costs involved.
Planar technologies may be utilized when the demand is less than one billion cells per annum, which can significantly reduce the costs of the process and, in turn, make these treatments available to patients at a lower price.
The utilization of autologous stem cell transplantation also offers an opportunity for patients to undergo stem cell therapy when a sibling does not provide an ideal match. Cells can be cultivated from the bone marrow and treated with specific drugs in order to yield targeted results. In the case of malignant cells in the body, bone marrow can be treated with monoclonal antibodies or cytotoxic drugs to target such cells when transplanted back into the patient.
Appropriate storage solutions are also taken into consideration to assist with the cultivation process, and to reduce the risk of enzymatic treatment leads to the differentiation of cells into other types. The culture condition becomes crucial for the expansion of stem cells. A PA6 conditioned medium, along with an enclosure of semi-permeable hollow fiber membranes, is one example of how the laboratory can keep certain stem cells from differentiating, while also assuring successful cultivation and expansion.
The GSCG’s Role In The New FDA Warnings
The fact that the FDA has announced the warnings against stem cell therapy products and had even taken action against a relatively popular company in the United States have now caused a limitation in access to these treatments. With a seizure of the services provided by companies that have been affected by the FDA’s actions and the knowledge of the misleading marketing information that was used in the campaigns initiated by these companies, patients might not be sure where to turn to right now.
The same problem is faced by many doctors in the field of regenerative treatments and medicine. Doctors in these fields need to ensure a facility they utilize to allow patients to undergo a treatment like a cell therapy can offer a safe procedure.
This is where the GSCG comes into the bigger picture. With the establishment of a permanent presence in Mexico, doctors are now given access to a facility that has an established history for providing quality stem cell therapy services to patients with qualifying diseases. The company is not only trusted but have been performing stem cell therapy procedures on patients that had led to successful results.
These treatments may help to assist in repairing neurological problems caused by disorders like Parkinson’s disease, stroke, or an injury to the spinal cord. They may also offer new hope to people with diabetes, heart disease, and even those who had previously suffered damage to their cardiovascular system due to a heart attack.
Conclusion
Even though recent publications from the FDA has caused concerns to be raised regarding stem cell therapy, many patients are still interested in undergoing these treatment procedures. A significant number of studies have provided evidence on the efficacy of treating tissue damage caused by various conditions through stem cell therapy. Patients interested in undergoing stem cell treatment are now advised to turn their interest to Stem Cells Centers in Cancun and related regions, where highly specialized and experienced doctors are able to provide a professional service.
- Published in Corporate News / Blog
EXOSOMES
One major aims of regenerative medicine is to replace lost tissue with new cellular material or to improve the regeneration of damaged, malfunctioning, diseases tissue and organs using stem cell transplantation. In view of this, the discovery of Extracellular vesicles (EVs) in the twentieth century have being considered as significant factors in inflammation and immune responses, antigen presentation, immunomodulation, coagulation, tissue regeneration, organ repair, cell-cell communication, senescence, proliferation and differentiation in the body [1]. Extracellular vesicles are believed to be involved in many biological processes and they can be modified to contain specific proteins, genetic lipids, and genetic materials including messenger RNA (mRNA), microRNA (mRNA), and other small non-coding RNAs, and genomic DNA (gDNA) from their progenitor cell.
Extracellular vesicles are classified into two groups which include; exosomes and ectosomes [2]. Exosomes are characterized with cup-shaped morphology, appearred as flattened spheres with diameters ranging from 30 to 150 nm. Similarly, exosomes have a characteristic lipid bilayer which has an average thickness of ∼5 nm. [3]. Thus, the lipid components of exosomes include ceramide (sometimes used to differentiate exosomes from lysosomes), cholesterol, sphingolipids, and phosphoglycerides with long and saturated fatty-acyl chains. The exosome is formed or derived from a multivesicular body (MVB) when the MVB fuses with the plasma membrane and is released into the extracellular environment.
Diagrammatic representation of a medium size Exosome.
Exosomes contain many types of biomolecule, including proteins, carbohydrates, lipids and nucleic acids which vary depending on the EV’s origin, its physiological and pathological state, and even the precise cellular release site. Thus, the protein composition within can also mark the existence of disease pathologies such as inflammatory diseases; however, exosomes also contain a number of common proteins as well as those that participate in vesicle formation and trafficking. Furthernore, exosome plays a role in intercellular communication by carrying proteins and RNAs between neighboring cells or even to distant organs, they also bind to cell membranes through receptor– ligand interaction and mediate antigen presentation, cancer progression etc.
Various techniques have being developed for the isolation of exosomes which includes; ultracentrifugation-based isolation techniques, size-based techniques, precipitation techniques, immune-affinity capture-based techniques, and some novel combination techniques [4]. Exosomes primarily exist in pellets after centrifugation 100000–200000×g and the use of Ultracentrifugation and ultrafiltration can be used to obtain purified exosomes in the laboratory, but this technology is difficult to apply on a large scale [5].
Exosomes can be stored at 4°C for up to 1 week for short-term while it can be stored at -80°C for long-term by suspending it in phosphate buffered saline.
More importantly, growing evidence has also suggested that exosomes play a key role in facilitating tumorigenesis by regulating angiogenesis, immunity, and metastasis. Circulating exosomes in body fluids and blood in particular are potentially non-invasive or minimally invasive biomarkers for early diagnosis and prognosis of various types of cancer.[6] As the first step towards improving human health, exosomes have to be reliably and efficiently isolated from complex biological matrices like blood, urine, and cerebrospinal fluid since they are currently tested as next-generation biomarkers in those body fluids.
In conclusion, it is advantageous to use exosomes for cell based treatments because the use of exosomes can avoid problems associated with the transfer of cells, which may already have damaged or mutated DNA [7]. Also, exosomes are small and can easily circulate through capillaries, whereas the cells used in other cell-based therapies, such as MSCs, are too large to go through capillaries, and thus cannot get beyond first pass capillary beds, such as the lungs.
The level of MSCs in cell-based therapies may quickly diminish after transplant while, exosomes can achieve a higher “dose” than the transplanted MSCs [8]. Similarly, exosomes can also be utilized to tackle toxicity and immunogenicity problems resulting from such biomaterial treatments as nanoparticles [9].
CLINICAL APPLICATION
Ø Immuno-modulatory and anti-inflammatory properties of Exosomes could be the potential biological mechanisms for clinical treatment to promote bone regeneration. [10]
Ø Adipose-derived stem cell-derived exosomes promote fracture healing in animals by binding to polylactic acid-glycolic acid scaffolds.
Ø Exosome can be used for the treatment of chronic kidney disease, type 1 diabetes mellitus, and skin damage [11].
Ø MSCs-derived exosomes have shown therapeutical benefits in stroke and intravenous administration of MSCs-derived exosomes induced an increase of neurogenesis, neurite remodeling, and angiogenesis. [12]
Ø Administration of MSCs-derived exosomes’ has being observed in a traumatic brain injury model by showing an inflammation reduction and good outcomes.[13]
Ø The injection of MSCs-derived exosomes has also been shown to be a possible treatment for spinal cord injury (SCI), by reducing inflammation and by promoting neuro-regeneration in rats after injury [14].
Ø Exosomes can be used as a delivery system of therapeutic signals or drugs due to their low immunogenicity, ability to cross the blood-brain barrier (BBB), and long half-life in circulation.
- Published in Corporate News / Blog
MESENCHYMAL STEM CELLS
Over the last decade, cellular therapy has developed quickly at the level of in vitro and in vivo preclinical research and in clinical trials. Thus, one of the types of adult stem cells that have provided a great amount of interest in the field of regenerative medicine due to their unique biological properties is Mesenchymal stem cells (MSCs).
Mesenchymal stem cells (MSCs) are known to be multipotent stromal cells that can differentiate into a variety of cell types which include osteoblasts (bone cells), chondrocytes (cartilage cells), myocytes (muscle cells) and adipocytes (fat cells which give rise to marrow adipose tissue). Furthermore, MSCs are responsible for tissue repair, growth, wound healing and cell substitution resulting from physiological or pathological causes; they have various therapeutic applications such as in the treatment of central nervous system afflictions like spinal cord lesions (1). Similarly, they are characterized by an extensive capacity for self-renewal, proliferation, potential to differentiate into multiple lineages and their immune-modulatory role on various cells.
Mesenchymal stem cells have the ability to expand in many folds in culture while retaining their growth and multilineage potential. Also, they can be identified by the expression of many molecules including CD105 (SH2) CD73 and CD34, CD45. Thus, these properties of MSCs make these cells potentially ideal candidates for tissue technology.
In addition, it has been discovered that MSCs, when transplanted systemically, have the ability to transport to sites of physical harm or damage in animals, suggesting that MSCs have migratory capacity. This migration property of MSCs is important in regenerative medicine, where various injection routes are utilized depending on the damaged tissue or organ.
The first source of Mesenchymal stem cells was in the bone marrow and considered to be the gold standard for clinical research, although various other sources have being discovered which include: Adipose tissue, Dental pulp, Mobilised Peripheral blood, Amniotic fluid, Joint synovium, synovial fluid, Endosteum, Periosteum, Menstrual blood and birth-derived tissues.
Cohnheim (German Biologist) hypothesized in the late nineteenth century that fibroblastic cells derived from bone marrow were involved in wound healing throughout the body, while in 1970, Alexander Friedenstein described a population of plastic-adherent cells that emerged from long-term cultures of bone marrow and other blood-forming organs, and he showed to have colony forming capacity and osteogenic differentiation characteristics in vitro as well as in vivo upon re-transplantation.
Arnold Caplan (1991), coined the term “mesenchymal stem cell and stated that the cells as multipotent mesenchymal cell populations which can differentiate into several tissue types, and demonstrated roles for MSCs in the regeneration of bone, cartilage or ligaments in animal and clinical studies. However, the first clinical trials of MSCs were completed in 1995 when a group of 15 patients were injected with cultured MSCs to test the safety of the treatment.
According to International Society for Cellular Therapy, the proposed minimum criteria to define MSCs include the following:
(a) The cells should exhibits plastic adherence
(b) The cell should possess specific set of cell surface markers, i.e. cluster of differentiation (CD) 73, D90, CD105 and lack expression of CD14, CD34, CD45 and human leucocyte antigen-DR (HLA-DR).
(c) The cells should have the ability to differentiate in vitro into adipocyte, chondrocyte and osteoblast.
Thus, these characteristics are valid for all MSCs, although few differences exist in MSCs isolated from various tissue origins.
Mesenchymal stem/stromal cells (MSCs) can be isolated from neonatal tissues, most of which are discarded after birth, including placental tissues, fetal membranes, umbilical cord, and amniotic fluid. Placenta is an ideal starting material for the large-scale manufacture of multiple cell doses of allogeneic MSC. The placenta is a fetomaternal organ from which either fetal or maternal tissue can be isolated.
MSC derived from placenta have long-term proliferation and immunomodulatory capacity, superior to bone marrow-derived MSC. The placenta is a fetomaternal organ consisting of both fetal and maternal tissue, and thus MSC of fetal or maternal origin can be, theoretically isolated. Thus, neonatal tissues are easily available and they have biological advantages in comparison to adult sources that make them a useful source for stem cells including MSCs. They appear to be more primitive and have greater multipotentiality than their adult counterparts.
Clinical Application
Ø MSCs have been widely used to treat immune-based disorders, such as Crohn’s disease, rheumatoid arthritis, diabetes, and multiple sclerosis.
Ø MSCs have been widely used as a treatment for numerous orthopedic diseases, including bone defects, osteoarthritis (OA), femoral head necrosis, degenerative disc, spinal cord injury, knee varus, osteogenesis imperfecta, and other systemic bone diseases.
Ø MSCs are promising cell source for treatment of autoimmune, degenerative and inflammatory diseases due to the homing ability, multilineage potential, secretion of anti-inflammatory molecules and immunoregulatory effects.
Ø MSCs play a key role in the maintenance of bone marrow homeostasis and regulate the maturation of both hematopoietic and non-hematopoietic cells.
Ø MSCs have been shown to be powerful tools in gene therapies, and can be effectively transduced with viral vectors containing a therapeutic gene, as well as with cDNA for specific proteins, expression of which is desired in a patient.
Ø It has been proved that MSCs can differentiate into insulin producing cells and have the capacity to regulate the immunomodulatory effects
REFERENCES
- Wei X, Yang X, Han ZP, Qu FF, Shao L, Shi YF. Mesenchymal stem cells: a new trend for cell therapy. Acta Pharmacol Sin. 2013 Jun; 34(6):747-54
- Friedenstein AJ, Piatetzky II S, Petrakova KV. Osteogenesis in transplants of bone marrow cells. J Embryol Exp Morphol. 1966;16:381–90
- Friedenstein AJ, Chailakhyan RK, Latsinik NV, Panasyuk AF, Keiliss-Borok IV. Stromal cells responsible for transferring the microenvironment of the hemopoietic tissues. Cloning in vitro and retransplantation in vivo. Transplantation. 1974;17:331–40.
- Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284:143–7.
- Caplan AI, Dennis JE. Mesenchymal stem cells as trophic mediators. J Cell Biochem. 2006;98:1076–84.
- Wang S, Qu X, Zhao RC. Clinical applications of mesenchymal stem cells. Journal ofHematology & Oncology. Apr 30, 2012;5:19.
- Dominici, M., Le Blanc, K., Mueller, I., Slaper-Cortenbach, I., Marini, F., Krause, D., Deans, R., Keating, A., Prockop, D. and Horwitz, E. (2006) Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular. Therapy position statement. Cytotherapy 8, 315–317
- Published in Corporate News / Blog
What Are The Best Options For Managing Chronic Pain?
Chronic pain. Roughly translated as a condition of persistent misery for people. It is a serious health condition that has both physical and psychological suffering and is often associated with a specific ailment like arthritis, migraine, frozen limb, etc. People who suffer from chronic pain perceive themselves to be in a state of constant agony and distress, which can affect their ability to lead a ‘normal’ life, from being able to go to work every day to social interaction.
There are several treatment options that people resort to for managing persistent pain conditions. In this article, we talk about those treatment options with the stem cell regeneration being the most current and promising one. Also, we will discuss the potential benefits and pros and cons of regenerative medicine.
What is chronic pain and how is it caused?
During ‘PAINWeek2017’ in Las Vegas, Dr. Jay Joshi, MD, explained the connection between inflammation and pain. He states that inflammation is triggered by various chemical and physical stimuli, and is a normal phenomenon of the body to protect the injured area and speed up the healing process. When inflammation exceeds its normal extent, it conversely delays the healing process and forms this cycle of non-resolving inflammation that leads to chronic pain.
Chronic pain is a multifactorial condition. It can be caused by medical harm, injury or nerve damage. However, it can also be caused by environmental factors, for example, back pain due to incorrect posture, or physiological factors such as aging, weak bones.
Common conditions that are related to chronic pain are:
- Osteoarthritis: the degeneration of cartilage in the joint and the corresponding bones, leading to pain and stiffness mainly in thumb, knee and hip joints.
- Migraine: a half-sided recurrent headache that gives a throbbing sensation and is accompanied by blurred vision and nausea.
- Multiple Sclerosis: a chronic, progressive condition that involves nerve cell sheath damage in the spinal cord and brain. Its symptoms include severe fatigue, speech disturbances, blurred vision and numbness.
- Neuropathy: a condition that involves damage to the peripheral nerves causing weakness, pain and numbness, particularly in the hands and feet.
- Fibromyalgia: a soft tissue disorder that involves widespread musculoskeletal pain also characterized by fatigue, sleep disturbance and mood swings.
According to a 2016 analysis by the National Health Interview Survey, approximately 50 million Americans suffer from chronic pain. It is considered to be one of the most common conditions that medical treatment is sought out for. Some of the repercussions of chronic pain that agonize people are:
- Depression and mood swings
- Poor performance
- Poor social life
- Restricted mobility
- Anxiety
- Poor self-perception about life
- Dependence on opioids
Although it is necessary to diagnose and treat the underlying cause of pain, doctors deal with pain as a separate entity and provide symptomatic treatments to save patients from the continuous discomfort it brings.
Management of Chronic Pain
Some of the common management strategies adopted by doctors are:
Analgesics
There are several over the counter pain killers available that are widely used by patients with chronic pain. These include paracetamol, acetaminophen, non-steroidal anti-inflammatory drugs (NSAIDs), aspirin, ibuprofen, naproxen, etc.
Although analgesics are considered safe, long term usage can produce a number of side effects. It can lead to liver damage and is specifically dangerous for patients with liver cirrhosis. Likewise, NSAIDs are known to affect the kidneys and increase the risk of a heart attack.
Anticonvulsants
Drugs that are originally used to treat epilepsy can also be an effective management strategy against chronic pain. For example, the second-generation anticonvulsants Gabapentin and Lyrica are approved by FDA for the treatment of pain. First-generation anticonvulsants such as carbamazepine and phenytoin are also considered effective. However, these first-generation drugs have more side effects when compared to second-generation drugs. They can cause side effects like ataxia, liver damage, sedation, etc.
Antidepressants
Doctors also prescribe antidepressants for the treatment of pain. These comprise of tricyclics like amitriptyline, imipramine or doxepin. These drugs can be given to patients with or without depression as it has distinct effects of pain relief and mood elevation.
Similarly, pain in fibromyalgia and diabetic nerve pain are treated with other types of antidepressants – serotonin and norepinephrine reuptake inhibitors (SNRIs) such as venlafaxine and duloxetine. These are equally effective as tricyclics but have comparatively milder side effects.
Opioids
Opioids such as codeine, morphine, oxycodone, etc. are very effective against severe chronic pain conditions. However, these drugs are not available over the counter and not provided by the pharmacist without prescription because of their risk of addiction.
Nonetheless, experts state that if the dose is vigilantly managed, their potential benefits can outweigh the risks. That is why doctors carefully observe patients prescribed with opioids for chronic pain to avoid any untoward dependence.
Alternative medicine
If traditional medicine hasn’t worked, many people often resort to other naturally occurring remedies for its cure. Alternative medicine varies from herbal remedies and therapies that are naturally available in plants that have curative properties. Cannabinoids, Akuamma and Kratom plants are a few examples with such characteristics that are currently trending.
Could Stem Cell Regeneration (SCR) be the answer?
As the human central nervous system matures and becomes more complex, the regenerative ability of the body further reduces. Regenerative medicine caters for this by focusing on the 3 Rs; Replacement, Repair, and Restoration of tissues and their function. This can be directed for the treatment of non-resolving inflammation by generating ex vitro cells, tissues or organs which can then be transplanted into the body.
Stem cell therapy is a unique model of treatment that replaces injured or lost cells, which can help 1.5 billion people that are experiencing chronic pain worldwide, with 23-26% of the people suffering from lower backache alone. According to Dr. Jay in ‘PAINWeek2017’, there are two types of stem cell regenerative therapy: Autologous and Non-Autologous.
In autologous SCR, mesenchymal stem cells (MSCs) are derived from adipose tissue of the patient and transplanted back into the injured area, where they potentially differentiate into the respective cells. For the other type, the MSCs are derived from embryonic tissues.
Research published in Pain Physician Journal in 2017 shows that MSC therapy is a great alternative because they are readily available. In addition, their homing potential is also considered a pro and they can be grown using standard culture techniques. It has also shown that autologous SCR is potentially safe for both humans and animals.
There has been a reported reduction from pre to post-operative pain with the use of SCR. However, various complications have been acknowledged in studies. Firstly, there’s a risk of tumor formation at the site of transplantation. Secondly, there have been instances of undesired bone formation. Furthermore, there is also a risk of abnormal immune reactions to occur.
The effective results of cell-based therapy are promising, with evident results in the treatment of chronic pain conditions in osteoarthritis, degenerative disc disease as well as neuropathic pain. However, there is still a need for advanced clinical research to deepen the understanding of the mode of action of MSCs and their potential outcomes inside the human body.
- Published in Corporate News / Blog
Which are the best options For Treating Common Sports Injuries?
As physicians, we are constantly dealing with sports injuries. While they are typically not seen as life-threatening with high chances of recovery, they could potentially cause further problems down the line for the patient. Sports injuries can also take months to heal and getting patients back on their feet can take a lot of time, effort and money, which can be frustrating for patients who want to get back to playing football every weekend or for those who have a skiing holiday booked in the next month.
Recently, there has been a massive surge in the use of stem cells as an alternative treatment to common sports injuries. This article aims to outline the benefits and risks attached to using stem cells and how this alternative treatment may help patients who are suffering from common sports injuries.
What Are The Most Common Sports Injuries?
We are constantly treating sports injuries on a regular basis. Some of the most common sports injuries doctors typically deal with are:
Ankle Sprain
An ankle sprain occurs when the ligaments in the ankle are stretched and eventually torn due to twisting or falling onto the foot. Ankle sprains are common among athletes and sports enthusiasts, however, if left untreated, the ankle can weaken, making it more vulnerable to further damage. This leads to long term problems such as chronic ankle pain, arthritis, and ongoing instability.
A sprained ankle can be easily diagnosed when we see displays of swelling, bruising, stiffness and pain when we attempt to touch or move the ankle.
Groin Pull
A groin pull is basically a groin strain. Strains occur when the muscles are overstretched, moving in directions that are not normal for them or pulled too forcefully or suddenly. This leaves them torn and damaged and results in tenderness and bruising in the groin and inner thigh area. This injury is common in athletes that play sports which require a lot of quick side-to-side movements.
A groin pull can be easily diagnosed after a thorough physical examination of the symptoms and possible tests such as x-rays and MRI’s.
Hamstring Strain
A hamstring strain occurs when the three muscles in the back of the thigh are overstretched from movements such as hurdling and kicking the leg out sharply. As these muscles are naturally tight and susceptible to sprains, it can take from six to twelve months to heal and are vulnerable to recurring injuries. Poor or lack of stretching are the likely causes of a pulled hamstring.
Shin Splints
Shin Splints is an inflammation of the muscles in the lower leg when they are overworked and stressed. Shin splints are often found in athletes that engage in sports that require a lot of running, dodging or quick stops and starts.
Knee injury: ACL tear
An ACL knee injury is a tear or sprain of the ligament that holds the leg bone to the knee. Sudden stops or changes in direction can tear the ACL and make that dreaded “pop” sound. Almost immediately, the knee will swell, feel unstable and be too painful to bear weight. This injury is common in athletes that engage in sports such as soccer, basketball, and downhill skiing.
ACL tears are commonly seen as a severe sports injury and can be traditionally treated with rehabilitation programs and surgery to strengthen or completely replace the torn ligament.
Knee injury: Patellofemoral Syndrome
Patellofemoral Syndrome occurs when repetitive movements of the kneecap (patella) are made against the thigh bone (femur) which damages the tissue. This knee pain is common among young adults and can be caused by a number of factors such as weakness in the thigh or buttock muscles, tight hamstrings, short ligaments around the kneecap or alignment problems through the feet.
Tennis Elbow (epicondylitis)
Tennis Elbow is common for athletes that play sports such as tennis or golf that require the player to ‘grip’ tightly and repetitively for an extended amount of time. This results in the ligaments of the forearm becoming strained and inflamed, making it painful to make wrist or hand motions.
What Are The Traditional Treatment Options & Management Strategies For Sports Injuries?
Traditional treatment methods and strategies for treating mild sports injuries such as sprains and strains can be done at home. Traditional advice given by doctors can include taking rest, applying ice to reduce the swelling and dressing the injury with compression bandages to support and assist healing. Nonsteroidal anti-inflammatory drugs (NSAIDs) such as ibuprofen may also be prescribed to help reduce swelling and to relieve pain.
Moderate severity injuries may be traditionally treated with immobilization strategies such as the use of crutches or a cast brace to assist the ankle to heal for the first few weeks. This may be followed with rehabilitation exercises to strengthen the ankle and prevent stiffness and future problems.
Severe sports injuries such as knee injuries are traditionally treated with surgery or rehabilitation programs to replace or restore the torn ligaments or muscles. This also goes for those patients who experience persistent problems after months of nonsurgical treatment.
Whatever the severity level of the sports injury, the healing process can take up to several months before the muscle restores to its natural condition. Injuries can also leave muscles and ligaments permanently weakened and susceptible to further injury.
Is Stem Cell Therapy An Option For Treating Sports Injuries?
The use of stem cells to treat sports injuries are becoming more popular due to its ability to grow new blood vessels that facilitate faster and better healing, decrease or prevent inflammation and release proteins (cytokines) that can slow down tissue degeneration and reduce pain. Sports injuries that include damage to tendons, ligaments, muscles, and cartilage are reported to be seeing the best results from stem cell therapy.
Dr. Bill Johnson, MD of Innovations Medical sees stem cell therapy as a revolutionary alternative to painful surgery and long recovery and states that,
“Patients who undergo stem cell therapy for their sports injury report a reduction in their painful symptoms and increase range of motion and increased mobility. Stem cell therapy helps to quickly reduce joint inflammation, and many patients see improvements in 1 to 2 days. Anti-inflammatory results of the procedure can last for 2 to 3 months and many patients see a gradual improvement in their condition over time.”
Even celebrity athletes such as Peyton Manning and Ryan Tannehill have used stem cell treatment in conjunction with traditional treatments with success. Stem cells are placed directly into the joint via surgical application or injection to aid quicker healing and promote the growth of cells needed to restore strength and flexibility in muscles and ligaments.
However, using stem cells as an alternative method to treat sports injuries is still a controversial subject and is very much still debated among medical professionals. Research is still undergoing to show whether or not stem cell therapy is the best solution. Critics of this therapy argue the fact that stem cell therapy doesn’t work any better than a placebo and that there is no clear evidence that this type of therapy is safe. Unwanted side effects can include swelling and pain and if stem cells are used from other sources or manipulated in any way, this can result in a higher risk of developing tumors.
- Published in Corporate News / Blog
How To Effectively Treat Amyotrophic Lateral Sclerosis (ALS)
Although a rare neurologic condition, Amyotrophic Lateral Sclerosis (ALS) is the most common type of Motor Neuron Disease (MND), a condition that affects the voluntary muscles. This is a progressive disorder that leads to muscle weakness and depletion due to nerve dysfunction.
ALS is also called Lou Gehrig’s disease, named after the football player who had this condition. The literal meaning of Amyotrophic is ‘no muscle nourishment’ which becomes the cause of muscle atrophy. ‘Lateral’ refers to the group of nerves in the spinal cord that sends signals to the muscles. It is these nerves that degenerate, leading to sclerosis in this region. In later stages, this affects the nerves that control breathing and hence can be fatal.
The initial symptoms of ALS include stiffness and muscle weakness, which gradually involves all the muscles under voluntary control. The affected regions and progressive pattern vary from one person to another. Some having difficulty holding a pen or a cup while others finding difficulty speaking, chewing or even talking. Thus, ALS is an ailment that affects daily life and makes simple tasks painful and troublesome.
According to the Center for Disease Control and Prevention (CDC), 14,500 to 15,000 people had ALS in the United States in 2016, with approximately 5000 people having a confirmed diagnosis for the condition annually. Although the average survival rate is three to five years, patients can live for ten years or more.
Are there different types of ALS?
Amyotrophic Lateral Sclerosis has two types:
Sporadic ALS: this is the most common one and affects 95% of sufferers. This type occurs without a clear cause.
Familial ALS (FALS): This type occurs in 5-10% of sufferers. This type of ALS is genetic and runs in families. This occurs due to abnormal changes to a gene that is then passed in generations.
What are the symptoms of ALS?
Early signs and symptoms might be unnoticeable and become perceptible after some time. Most clinical signs are evident of upper motor neuron and lower motor neuron lesion. The limb onset ALS (70%) involves initial symptoms in the limbs while the bulbar onset ALS (25%) is characterized by speech and swallowing problems. This is followed by weakness in the limbs later. The remaining 5% of the patients have respiratory involvement in the early period. [5]
Most common symptoms include:
- Muscle weakness in the limbs (distal or proximal)
- Asymmetric progressive muscle wasting
- Difficulty in motor activities like walking, talking, chewing
- Weakness in arms, legs, hands, and feet
- Muscle cramps and twitching
- Slurred speech
- Fatigue
- Emotional liability (episodes of uncontrolled laughing and crying)
- Difficulty in maintaining posture and gait
- Difficulty in breathing and swallowing
With the progression of the disease, symptoms may spread to all parts of the body. In some patients, frontotemporal dementia may occur resulting in poor memory and decision-making abilities.
What Causes ALS?
The exact cause of ALS has not been known by scientists to date. However, research is being carried out to understand what causes ALS. There are several different factors such as:
Genetic changes
Studies have shown that 5 to 10% of cases of ALS are caused by genetic mutations. For example, changes to the gene that makes SOD1 protein causes damage to motor neurons.
Environmental factors
No major association has been established between environmental factors like toxins, viruses, diet or physical trauma and the risk of development of ALS. However, there is ongoing research on the subject. Studies have shown that some athletes are at a higher risk of acquiring ALS due to vigorous physical activity.
Chemical disturbance
Glutamate is the neurotransmitter that is in control of signals to and from the brain. Accumulation of this neurotransmitter within the spaces surrounding the nerves damages them.
Research has also shown mitochondrial structural and functional abnormalities, as well as defects in axonal structure and transport, could be the causative agents for ALS.
How Do We Diagnosis ALS?
When it comes to diagnosis, there are no specific tests that can provide a definitive diagnosis for ALS. However, doctors conduct a series of tests to rule other similar diseases. A full medical history check and a neurologic examination are undertaken at regular intervals to assess the progressive worsening of symptoms.
Running the following Image testing diagnostic tests can help reach a diagnosis:
- Electromyography (EMG) – EMG records the electrical activity of the muscle fibers.
- Nerve Conduction Study (NCS) – NCS assesses the electrical activity of the nerves and muscles.
- Magnetic Resonance Imaging (MRI) – MRI rules out other possible conditions such as a tumor or cyst in the spinal cord, cervical spondylosis, or a hernia in the neck that could be causing the nerve compression.
Laboratory tests such as blood screening and urine tests can also be carried out so that other diseases can be eliminated.
What Are The Treatment Options & Management Strategies For ALS?
ALS is managed through a multidisciplinary approach.
Unfortunately, there is no definitive cure for the disease at this time. Management of ALS is done through symptomatic treatment to ease the condition of the patients and prevent unnecessary complications:
Support – Physicians, psychologists, speech therapists, nutritionists, and home care assistance all play a vital role in making life easier for patients with ALS.
Medication – Riluzole (Rilutek) and Edaravone (Radicava) are the drugs approved by the U.S Food and Drug Administration (FDA) for treating ALS. Riluzole is believed to reduce glutamate levels thereby, decreasing damage to the motor neurons. Edaravone acts an antioxidant and is believed to expel free radicals and reduce the oxidative stress in the motor neurons.
Lifestyle Habits – Physiotherapists can recommend exercise and physical activity like walking, swimming, and bicycling that may improve muscle strength and help elevate mood without overstressing the muscles.
Speech Therapy – therapists can help patients with ALS to employ strategies to speak clearly. They may also recommend computerized aids such as speech synthesizers and eye-tracking technology to help people learn ways for responding by nonverbal means.
Diet – Nutritionists may formulate a diet plan for patients, which consists of food that is easy to swallow and provides enough nourishment and calories for the patients to maintain adequate energy levels and to prevent excessive weight loss
Breathing Support – Patients with ALS may suffer from shortness of breath and difficulty breathing during physical activity or while lying down. If this is the case, doctors can recommend Non-Invasive Ventilation (NIV) that provides breathing support through the nose or mouth. NIV improves quality of life and increases the number of years of survival for patients.
Is Stem Cell Therapy An Option?
As previously mentioned, there is no curable treatment for ALS available. However, scientists are researching Stem Cell Therapy as the new favorable approach in the treatment of neurologic disorders.
There is a rising interest in Stem Cell Therapy as a promising remedy for curing ALS. Mesenchymal stem cells are particularly believed to be the most suitable ones due to their availability, absence of ethical issues and positive results in various experiments.
Studies and clinical trials have begun to apprehend the benefits of MSC transplantation. They demonstrate that MSCs lead to a partial recovery of motor neurons and a delay in disease progression. Also, there has been no evidence of a major adverse effect after MSC transplantation.
When testing this newfound research on animals, the lifespan of the subjected animal has increased with MSC transplantation. These positive results have encouraged the administration of MSC in ALS patients.
However, despite the safe outcomes of MSC transplantation in humans, results show that there is only a partial improvement in ALS sufferers with only a few cases that showed a delay in disease progression. Hence, there is a need for further studies and trials on a higher number of human subjects for a better understanding of MSC effects so that more significant conclusions can be reached.
- Published in Corporate News / Blog
Which Works Better, Viscosupplementation, or Platelet Rich Plasma?
One of the treatments that the FDA approved for knee arthritis is a Hyalronic Acid (HA) injection, sometimes also known as Viscosupplementation. It has been incredibly successful for knee arthritis. In fact, so successful that many physicians are starting to use it on other parts of the body, like the hip and shoulder, which the FDA does not approve of.
HA, when injected, works like the fluid that naturally surrounds your joints. This fluid can be like a lubricant for your joints, and absorb shock, allowing bones that otherwise cause arthritis pain cause much less. Over time, it is even absorbed into the joint, which can cause the body to create a more stable cartilage all on its own.
The evidence for this treatment is astounding, with a systematic review of 76 trials, all of which were randomized controller trials. The review noted that HA, when injected, can benefit function, reduce pain, and can be a reliable and effective treatment for knee osteoarthritis.
On the other hand, there is PRP therapy. Platelet Rich Plasma, or PRP, which is a type of blood that has 6 to 10 times more platelets than what is normally found in blood. They even contain many growth factors, such as Epidermal Growth Factor, Connective Tissue Growth Factor, and many more. These can help heal injured parts of the body by using the bodies natural healing tools.
However, PRP is not regulated by the FDA, and devices that are used to make PRP require said approval. Aside from this, multiple studies shoe that PRP can be very effective in the treatment of tendon injuries, as well as for osteoarthritis. This treatment can even help in the reduction of pain. There are even more studies being conducted on whether it can help other things, such as hair regrowth, cardiac muscle repair, and even dermatologic rejuvenation.
So should you use HA injections, or PRP?
In many studies, PRP has bee demonstrated to work just as well, if not better than HA. HA is also only FDA approved for the knee, meaning that it is not approved or covered for the use in any other joint. Also, the risk of infection and rejection is far less while using PRP, as it is a substance that comes from your own body, and contains white blood cells, which can help fight infection.
PRP also saves money in the long run, as using HA in a joint other than the knee is not FDA approved or covered by insurance. As a result, it can cost your patient 1500$ or more. This can even be on top of various other charges, such as doctors visits, and even the injection itself. PRP, on the other hand, only costs from 800 to 1200$ out of pocket.
So PRP has been demonstrated to be just as effective, if not better, than HA injections when it comes to arthritis pain. It does not pose a risk for infection or and auto-immune reaction either, and is even far cheaper than HA. So picking which one to use should be a no-brainer.
- Published in Corporate News / Blog